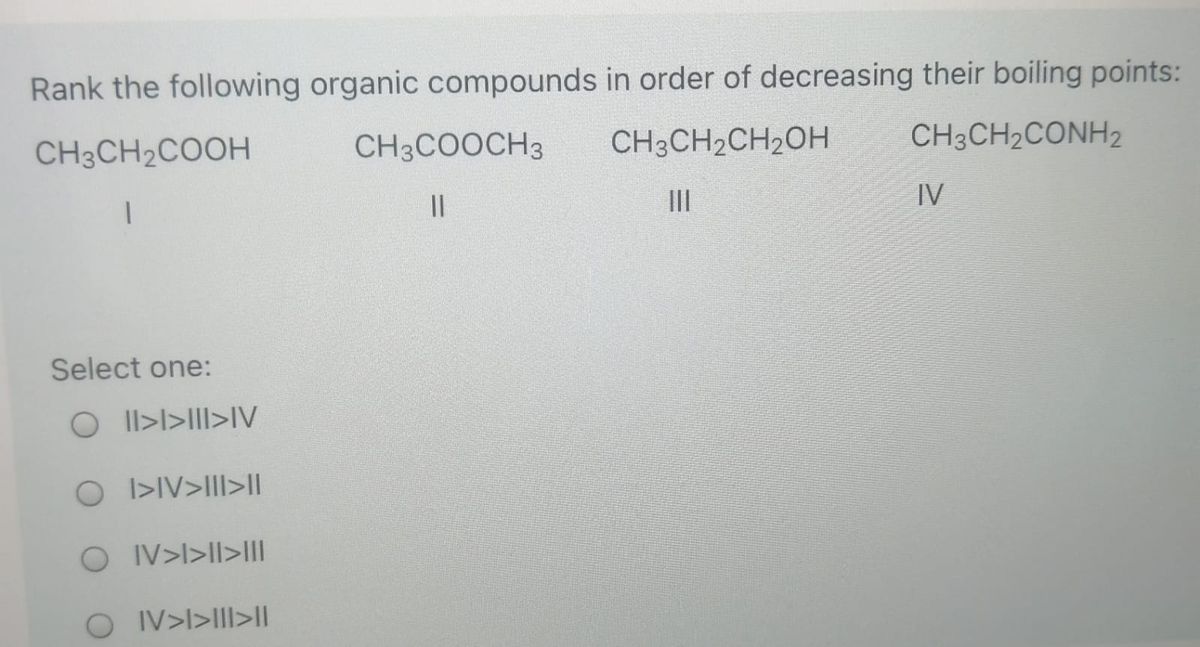
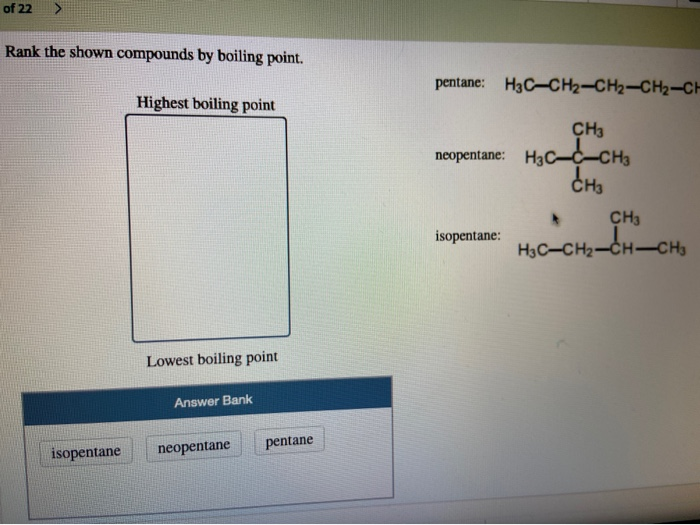
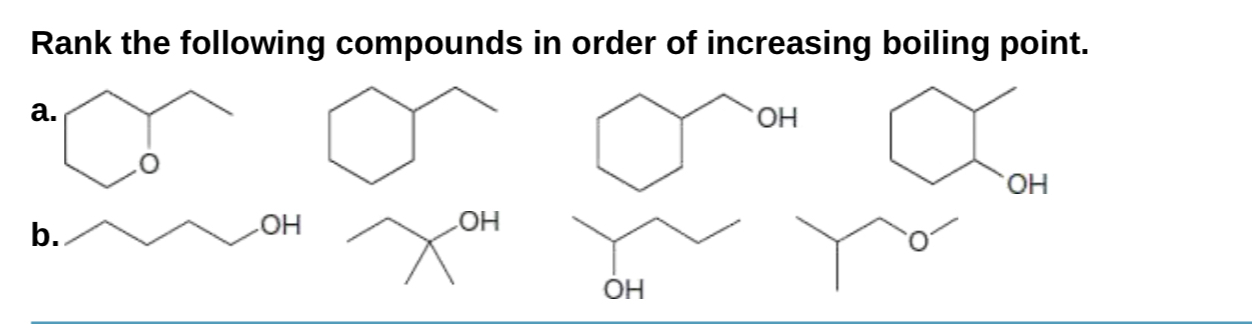
Rank The Compounds According To Their Boiling Point.

Ever wondered why some perfumes evaporate in a flash while others linger all day? Or why water boils at a much higher temperature than, say, rubbing alcohol? The answer lies in a fascinating property called boiling point! Understanding boiling points isn't just for chemistry nerds; it's surprisingly relevant to everyday life, from cooking to understanding the weather. So, let's dive into the fun game of ranking compounds based on their boiling points!
The purpose of ranking compounds by their boiling points is to predict their behavior under different temperatures. Will something be a gas, a liquid, or a solid at room temperature? Knowing its boiling point helps us figure that out! This knowledge is incredibly beneficial in a variety of fields. Chemists use it to separate and purify substances. Engineers use it to design efficient distillation processes. And even chefs implicitly use it when deciding the cooking temperature for different ingredients.
So, how do we actually go about ranking these compounds? It all boils down (pun intended!) to the strength of the intermolecular forces holding the molecules together. Think of these forces like tiny magnets holding hands between molecules. The stronger the "hand-holding," the more energy (heat) we need to break those bonds and allow the molecules to escape into the gas phase – hence, a higher boiling point.
Must Read
Here’s a simple ranking system, starting with the factors that lead to higher boiling points:
- Molecular Weight: Generally, the heavier the molecule, the higher the boiling point. Imagine trying to push a small car versus a large truck; the truck requires more force to move. Similarly, heavier molecules require more energy to enter the gas phase.
- Hydrogen Bonding: This is the superstar of intermolecular forces! Compounds with hydrogen bonds (like water, H₂O) have significantly higher boiling points. These bonds are extra strong attractions between molecules containing hydrogen bonded to highly electronegative atoms like oxygen, nitrogen, or fluorine.
- Dipole-Dipole Interactions: Molecules with permanent dipoles (uneven distribution of electron density, making one end slightly positive and the other slightly negative) also experience stronger attractions than nonpolar molecules. These "partial charges" attract each other, raising the boiling point.
- London Dispersion Forces: Even nonpolar molecules have temporary, fleeting dipoles that cause weak attractions. These forces, called London dispersion forces, are present in all molecules, but they are the only forces acting between nonpolar molecules. Their strength increases with the size and shape of the molecule; larger surface area leads to stronger attractions.
Let's consider an example. Imagine we have methane (CH₄), water (H₂O), and ethanol (CH₃CH₂OH). Methane is a small, nonpolar molecule with only London dispersion forces. Water is smaller than ethanol but has incredibly strong hydrogen bonds. Ethanol is larger than water and also has hydrogen bonds, but its nonpolar part is more significant, making the hydrogen bonding effect less dominant than in water.
Therefore, we can confidently predict the following boiling point ranking (lowest to highest): Methane, Ethanol, and Water. Methane boils at a chilly -161.5 °C, Ethanol boils at 78.37 °C, and Water boils at a familiar 100 °C. See how these rules play out in real life?
So next time you're waiting for water to boil or smelling your favorite fragrance, remember the fascinating world of intermolecular forces and the surprisingly useful art of ranking compounds by their boiling points!