Properties Acids And Bases
Okay, picture this: last week I’m attempting a culinary masterpiece (or at least, that's what I thought I was doing). I'm following this fancy recipe for lemon bars, and I, in my infinite wisdom, decided to "eyeball" the lemon juice. Let's just say the resulting puckery face my family made could curdle milk. It was… aggressively sour. Like, tongue-numbing sour. That, my friends, is a prime example of an acid doing its thing. But what is it doing, exactly? Let's dive in!
Acids: The Sour Bunch
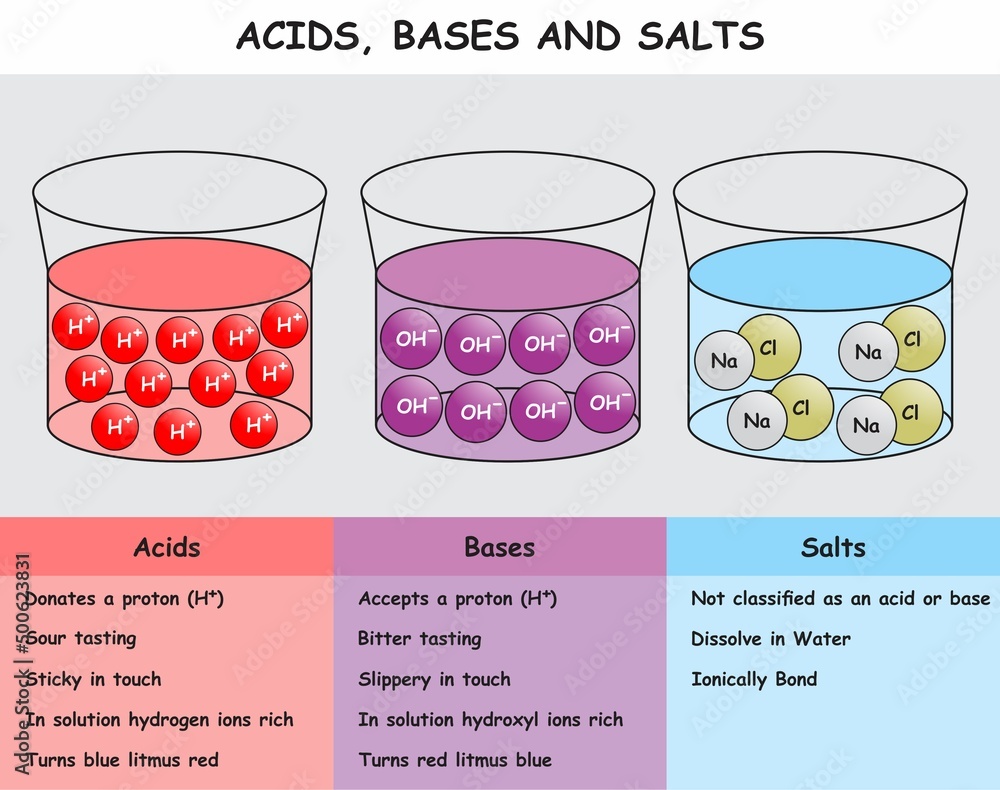
So, what makes something an acid? Well, on a fundamental level, acids are substances that can donate a proton (H+). Think of it like this: they’re always offering a hydrogen ion to the party. In water, this increased concentration of H+ ions is what gives acids their characteristic properties.
Taste: They taste sour. (Duh, lemon bars, remember?). Don't go around tasting random chemicals in your garage, though! Safety first! That lemon juice? Acetic acid (vinegar)? Ascorbic acid (vitamin C)? All acids, all sour.
Must Read
Reactivity: Acids are notorious for their reactivity. They can react with metals, carbonates, and bases (more on those later). Remember those baking soda volcanoes you made in elementary school? That’s an acid (vinegar) reacting with a base (baking soda) to produce carbon dioxide gas. Science is fun, isn’t it?
Corrosiveness: This is where things get serious. Strong acids can be corrosive, meaning they can damage or destroy other substances, including your skin (hence, the lab coats and safety goggles). Hydrochloric acid (HCl), for instance, is a strong acid used in cleaning and industrial processes. Handle with extreme care!

Bases: The Slippery Crew
Now, let’s flip the script and talk about bases. Where acids donate protons, bases accept protons. They’re the proton magnets of the chemical world.
Taste: Bases often taste bitter. Think of that soapy aftertaste you sometimes get from improperly cleaned dishes. (Ew, right?)
Feel: Bases feel slippery. Think of soap again. This is because they react with the oils on your skin to form soap-like substances. Kinda cool, kinda gross, all science!

Reactivity: Like acids, bases are reactive. They react with acids (duh!), and some can react with metals. Sodium hydroxide (NaOH), also known as lye, is a strong base used in drain cleaners and soap making. Again, super corrosive, super dangerous. Don’t mess around with this stuff without proper training!
Alkalinity: You might hear the term "alkaline" used interchangeably with "basic." It generally refers to bases that are dissolved in water. Alkaline water is having a moment, but that’s a topic for another time!
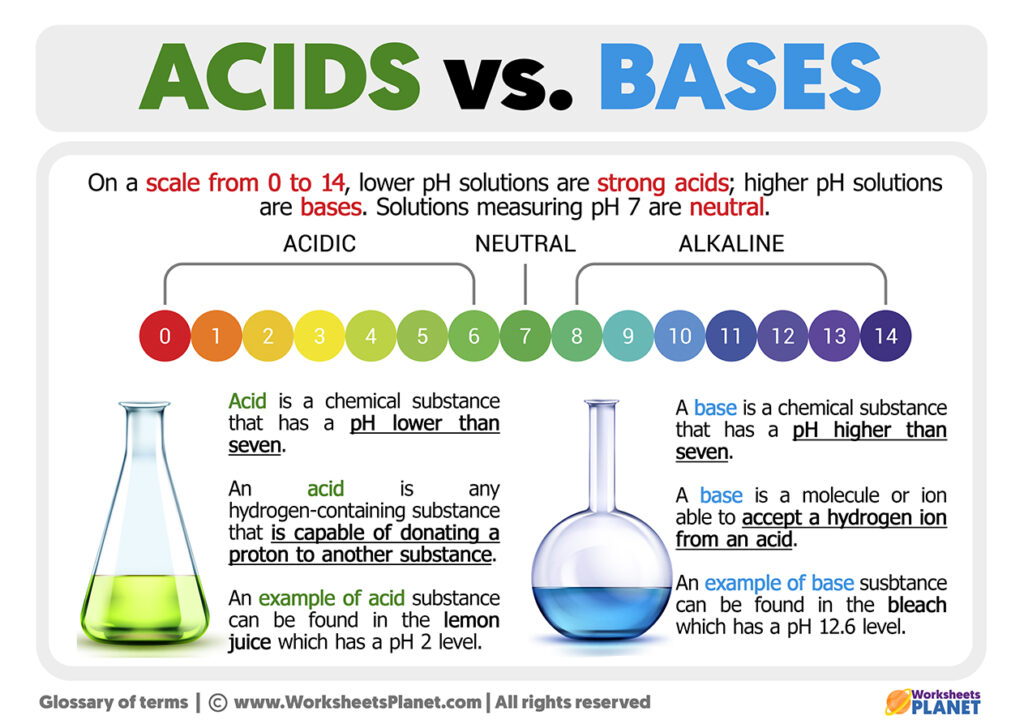
The pH Scale: Measuring Acid-Base-ness
So, how do we know how acidic or basic something is? Enter the pH scale! This handy-dandy scale ranges from 0 to 14. 7 is neutral (pure water), values below 7 are acidic, and values above 7 are basic. The further away from 7 you get, the stronger the acid or base.
For example:
- 0-6: Acidic
- 7: Neutral
- 8-14: Basic
Lemon juice is around pH 2 (highly acidic), baking soda solution is around pH 8 (slightly basic), and drain cleaner is around pH 14 (extremely basic). Knowing the pH of a substance is crucial for understanding its properties and potential hazards.

Acids and Bases: A Balancing Act
Acids and bases are everywhere, playing crucial roles in everything from digestion (your stomach acid is hydrochloric acid!) to cleaning your house (bases in cleaning products). They're fundamental to chemistry and biology. They even helped me learn a valuable lesson about measuring ingredients properly in lemon bars. (Okay, maybe I learned it, anyway.)
Understanding the properties of acids and bases is essential for safety, for cooking, and for just generally being a more informed human being. So next time you taste something sour or feel something slippery, remember the protons, remember the pH scale, and remember that chemistry is all around us, even in the most unexpected places.
Now if you'll excuse me, I'm going to go try to salvage those lemon bars... wish me luck!
