Potassium Permanganate Molar Mass
Okay, so you've stumbled upon potassium permanganate. Sounds kinda sci-fi, right? And maybe you've heard whispers of its molar mass. But what is that, and why should you even care? Let's break it down in a way that's less chemistry textbook and more chill chat over coffee.
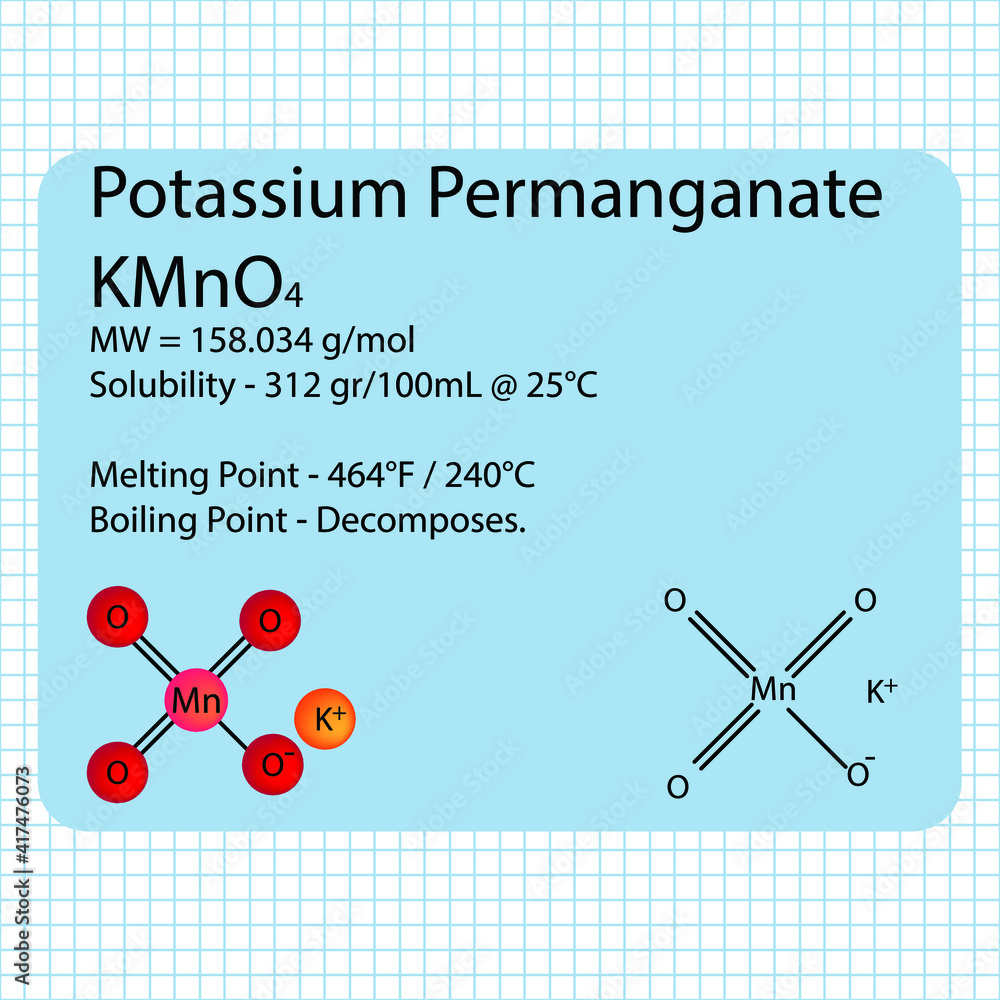
First things first: potassium permanganate. Chemically, it's KMnO4. Imagine it as a little molecular team with one potassium player, one manganese player, and four oxygen players. That beautiful purple hue? That's all thanks to this particular combination.
What's Molar Mass Anyway?
Now, for the molar mass. Think of it as the weight of one mole of these little teams. A mole, in chemistry terms, isn't the furry kind that digs in your garden. It's a specific, gigantic number – 6.022 x 1023 to be exact (also known as Avogadro's number!). Sounds intimidating, but basically, it's a standard "chemist's dozen."
Must Read
So, molar mass tells us how heavy a "chemist's dozen" of potassium permanganate molecules is. Why do we need it? Because it's super useful for all sorts of things, from calculating how much of something to use in an experiment to understanding how reactions work. It's like knowing the recipe measurements before you start baking – crucial for getting the results you want!
Think of it like this: If you wanted to make a huge batch of cookies, you wouldn't just eyeball the ingredients, right? You'd use a recipe to make sure you had the right amount of flour, sugar, and chocolate chips. Molar mass is like the recipe for chemical reactions, helping us get the quantities just right.

Finding the Molar Mass of KMnO4
Alright, time for some simple math. Don't worry, it's more like adding than calculus. To find the molar mass of potassium permanganate, we need to add up the atomic masses of each element in the compound.
Where do we find those atomic masses? Usually on the periodic table! You can find them rounded to two decimal places in most sources. Here's what we need:
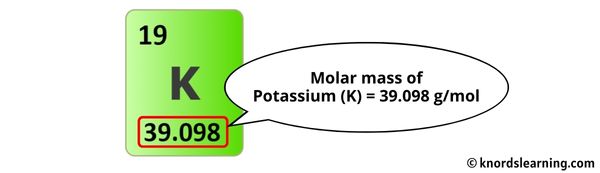
- Potassium (K): 39.10 g/mol
- Manganese (Mn): 54.94 g/mol
- Oxygen (O): 16.00 g/mol
Now, let’s plug it into our KMnO4 formula:
(1 x Potassium) + (1 x Manganese) + (4 x Oxygen) = Molar Mass of KMnO4
(1 x 39.10) + (1 x 54.94) + (4 x 16.00) = ?
39.10 + 54.94 + 64.00 = 158.04 g/mol

So, the molar mass of potassium permanganate is approximately 158.04 g/mol.
That means one mole (that giant "chemist's dozen") of KMnO4 weighs about 158.04 grams. Easy peasy, right?
Why is This Cool?
Okay, you might still be thinking, "So what?" But here's where it gets interesting. Potassium permanganate isn't just some random chemical. It's a powerful oxidizing agent, which basically means it loves to react with other stuff, especially things that are easily oxidized.
Think of it like this: Imagine potassium permanganate is a hungry Pac-Man, and other chemicals are dots. It gobbles them up, changing their chemical structure in the process. This oxidizing power makes it useful in a surprising number of ways:
- Water Treatment: It helps to disinfect water by oxidizing impurities.
- Photography: It's used in some developing processes.
- Medicine: Diluted solutions can be used as an antiseptic for minor skin conditions.
- Survival situations: It can be used to start fires! (Though please be careful!)
And because we know its molar mass, we can precisely control how much potassium permanganate we use in these applications. Want to disinfect a specific volume of water? Knowing the molar mass lets you calculate exactly how much KMnO4 to add for safe and effective treatment.
So, the next time you see that intriguing purple powder, remember its molar mass. It’s not just a number; it’s the key to unlocking its potential and understanding its fascinating chemistry. Plus, knowing fun facts about molar mass and chemistry is a fantastic way to impress your friends at parties… or at least spark some interesting conversations! Who knew chemistry could be so cool?
