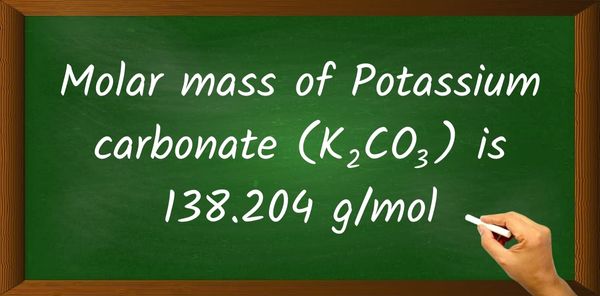Potassium Carbonate Molar Mass
Ever wondered what connects grandma's old-fashioned soap to the fluffy texture of your favorite baked goods? Or perhaps you've pondered how certain specialized glass is made to be so incredibly strong? The answer, in part, lies with a fascinating chemical compound called potassium carbonate. And understanding its molar mass is like unlocking a secret code to understanding how it works its magic. Don't worry, we're not diving into a complicated chemistry textbook. Instead, let's explore this seemingly obscure topic in a fun and engaging way!
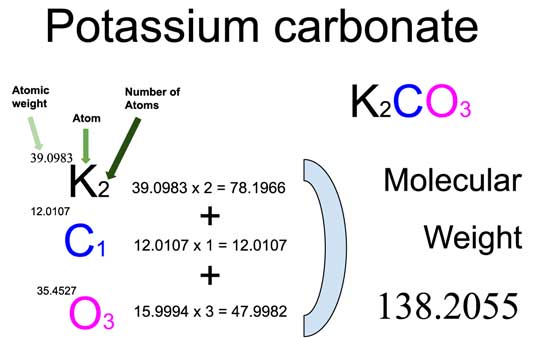
So, why should you care about potassium carbonate's molar mass? Well, molar mass is simply the mass of one mole (a specific number of atoms or molecules) of a substance. Knowing the molar mass allows scientists, and even amateur cooks and craftspeople, to accurately measure and use chemicals. In the case of potassium carbonate, it's crucial for ensuring the right proportions in various applications. Think of it like a recipe – too much or too little of an ingredient can drastically change the outcome.
Potassium carbonate (K₂CO₃) has a surprisingly wide range of uses. For centuries, it was a key ingredient in soap making. The potassium carbonate, often extracted from wood ashes, reacts with fats to create a soft, gentle soap. Even today, you might find it listed in the ingredients of specialty soaps. In the kitchen, it acts as a leavening agent, particularly in traditional German gingerbread and other dense baked goods. It creates a lighter, more airy texture. Beyond the home, potassium carbonate is crucial in the production of certain types of glass, like optical glass and TV screens, contributing to their clarity and strength.
Must Read
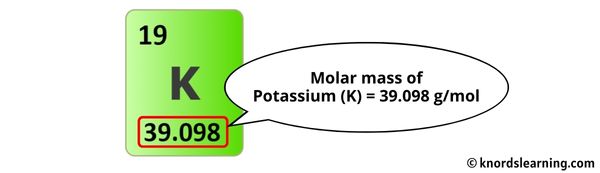
In education, understanding the molar mass of potassium carbonate is a fundamental concept in chemistry. Students learn to calculate it by adding up the atomic masses of each element in the compound (potassium, carbon, and oxygen), which helps them grasp the relationship between atoms, molecules, and mass. This knowledge then becomes a building block for more advanced chemical calculations and understanding of reactions.
You might be wondering how you can explore this further. While handling pure potassium carbonate requires proper safety precautions (like wearing gloves and eye protection), there are safer ways to engage with the concept. For example, you could research the history of soap making and how potassium carbonate was traditionally obtained. Or, if you're a baker, try experimenting with a recipe that calls for potassium carbonate (also known as potash) – just be sure to follow the recipe carefully. Even simpler, try finding products around your house that list potassium carbonate as an ingredient and see if you can deduce its role based on the product's function.
Understanding potassium carbonate's molar mass isn't just about memorizing numbers; it's about appreciating the science that's woven into the fabric of our everyday lives. From the soap we use to the screens we watch, this unassuming chemical compound plays a vital role, and its molar mass is the key to understanding its power.