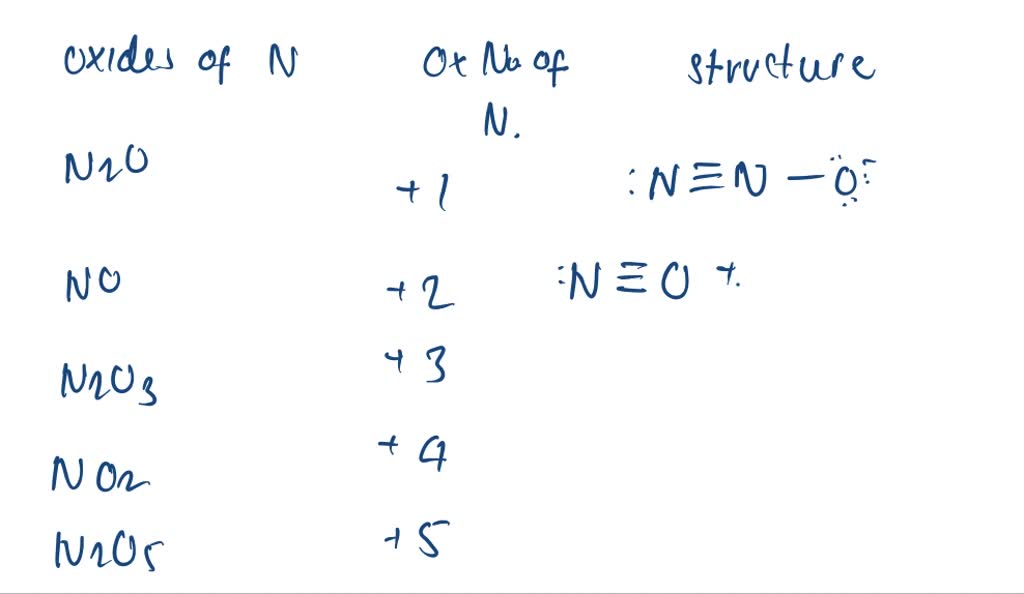Oxidation Number Of Nitrogen
Let's face it, diving into the world of chemistry can feel a bit like entering a secret society, complete with its own language and customs. But trust me, there are hidden treasures to be found! And one of the most surprisingly useful of these treasures is understanding the concept of oxidation numbers, especially when it comes to nitrogen. You might not think you'd use it every day, but knowing a little about nitrogen's oxidation states can unlock a whole new level of appreciation for the world around you.
So, why should you care? Well, the oxidation number of nitrogen essentially tells us how many electrons a nitrogen atom has gained or lost when it forms a chemical bond. It's like a chemical bookkeeping system, helping us understand how nitrogen interacts with other elements. Why is this important? Because nitrogen is a key component of so many things we rely on! Think about it: from the air we breathe to the food we eat, nitrogen is everywhere.
The benefits of understanding nitrogen's oxidation numbers are surprisingly practical. For example, it helps us understand the nitrogen cycle, a vital process that ensures a continuous supply of nitrogen for plant growth. Without nitrogen, plants can't produce proteins and other essential compounds, and therefore, we wouldn't have food! Furthermore, knowing about oxidation numbers allows us to predict the behavior of different nitrogen-containing compounds. This is crucial in agriculture, where fertilizers containing nitrogen are used to boost crop yields. Understanding the oxidation states of nitrogen helps us optimize fertilizer use, preventing over-fertilization and minimizing environmental impact.
Must Read
You've probably encountered nitrogen oxides (NOx) in discussions about air pollution. These compounds, formed during combustion processes like those in car engines, contribute to smog and acid rain. The different oxidation numbers of nitrogen in these oxides (e.g., +2 in NO, +4 in NO2) determine their reactivity and their environmental impact. Being able to identify these oxidation numbers gives you a deeper insight into the chemistry behind air quality.
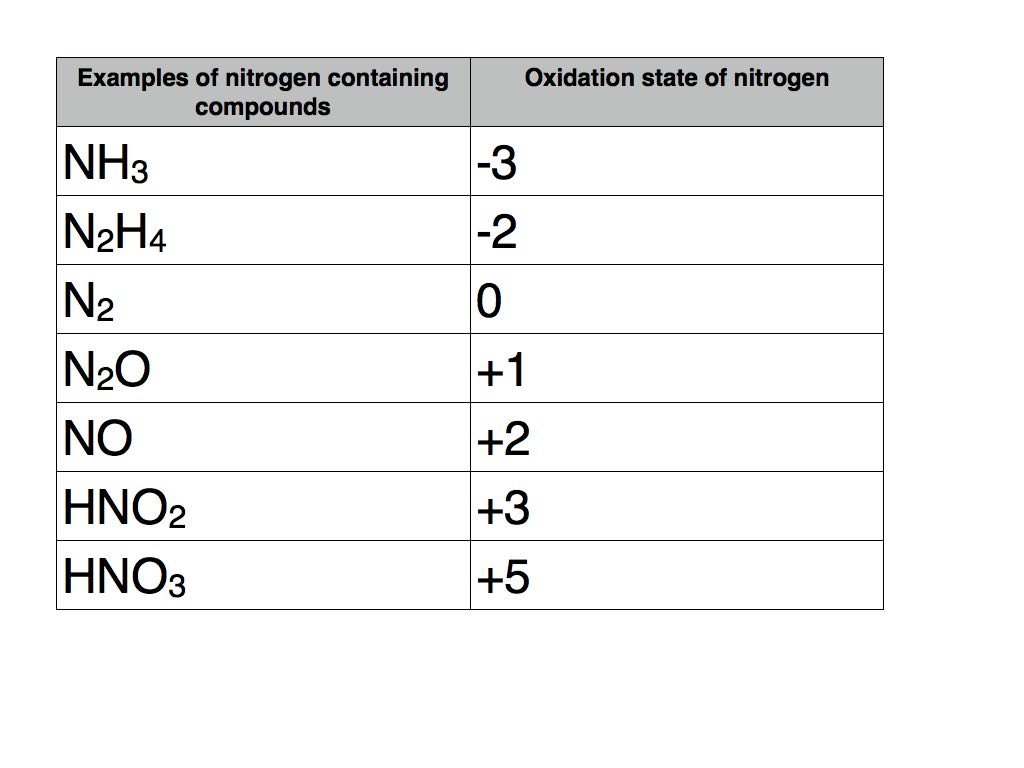
Here are some common examples of how nitrogen appears with different oxidation numbers: In ammonia (NH3), nitrogen has an oxidation number of -3. In atmospheric nitrogen (N2), it's 0. In nitrous oxide (N2O), or laughing gas, it's +1. In nitric acid (HNO3), it's +5. The range is quite remarkable! These variations dictate the substance's chemical behavior.

So, how can you enjoy this fascinating topic more effectively? First, start with the basics. Understand the rules for assigning oxidation numbers. There are plenty of online resources and textbooks that can guide you. Then, practice! Work through examples of different nitrogen-containing compounds and try to determine the oxidation number of nitrogen in each one. Don't be afraid to make mistakes; they are part of the learning process. Finally, try to connect the concept to real-world applications. Think about the nitrogen cycle, fertilizers, and air pollution. The more you can relate the concept to your everyday life, the more engaging and rewarding it will become.
Ultimately, understanding the oxidation number of nitrogen isn't just about memorizing rules; it's about gaining a deeper appreciation for the intricate chemistry that governs our world. It's a tool that allows us to understand and solve problems related to agriculture, the environment, and even human health. So, embrace the challenge and discover the power of nitrogen's oxidation states!