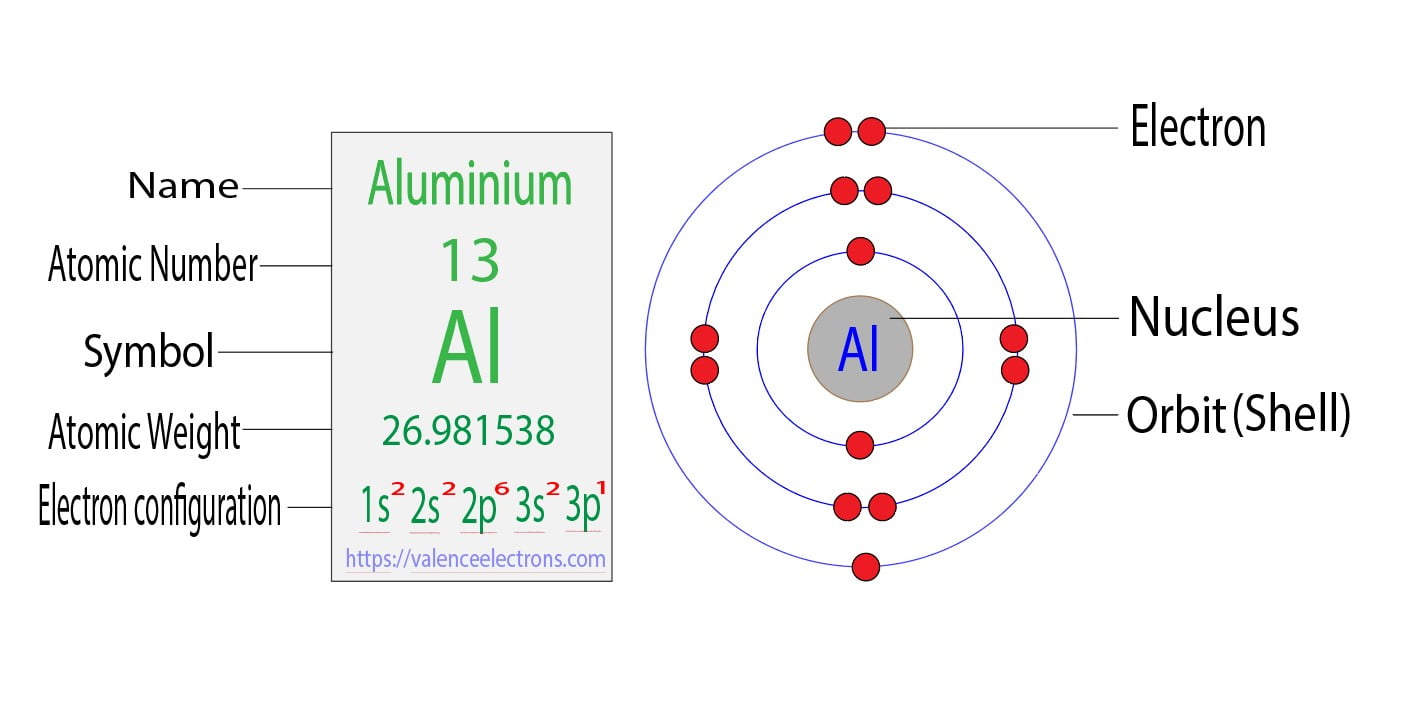
Orbital Diagram For Aluminum

Ever wonder what's buzzing inside that aluminum can of your favorite soda? We're not talking about the fizz, but something far tinier: its electrons! Let's peek at aluminum's orbital diagram - a sort of electron blueprint, without all the complicated jargon.
Level One: The Inner Circle
Imagine electrons as tiny, energetic kids playing musical chairs around the nucleus (the atom's core). The first energy level is like the front row of chairs, closest to the teacher (the nucleus). Only two electrons can squeeze into this tiny space, and they are snug as bugs in a rug.
This space is designated as the 1s orbital. We'd draw it as one box with two arrows inside, one pointing up and one pointing down. Think of the arrows as representing the electrons’ spin – tiny, internal rotations. These kids are always on the move!
Must Read
Level Two: Growing Room
Now, picture the next energy level, a slightly bigger circle with more space. Eight electrons can now join the party! Here, we have two types of "chairs": the 2s orbital and the 2p orbitals.
The 2s orbital is like a slightly bigger, comfier chair for two more electrons. Again, we draw one box and two arrows.
The 2p orbitals are where things get interesting. They are more like a set of three chairs, each holding two electrons, adding up to six in total. Think of it as a tiny electron apartment complex.
We draw the 2p orbitals as three boxes, each getting its two electrons (with opposite spins, of course). This completes the second energy level. Everyone is paired up, and happy.
Level Three: Aluminum's Unique Style
Here comes the star of our show: Aluminum! Aluminum has 13 electrons in total. This means we've already filled up the first two levels with 2 + 8 = 10 electrons.
Time for the third energy level! We start with the 3s orbital, which, you guessed it, holds two more electrons. This leaves us with just one electron to deal with.
Now for the fun part: the 3p orbitals. We have three boxes ready and waiting, but only one electron looking for a spot. It's like being the first person at a movie theater – so many choices!
Following Hund's Rule (yes, there's a rule!), this electron will happily occupy one of the 3p orbitals all by itself. No need to pair up until it absolutely has to! Think of it as declaring its independence.
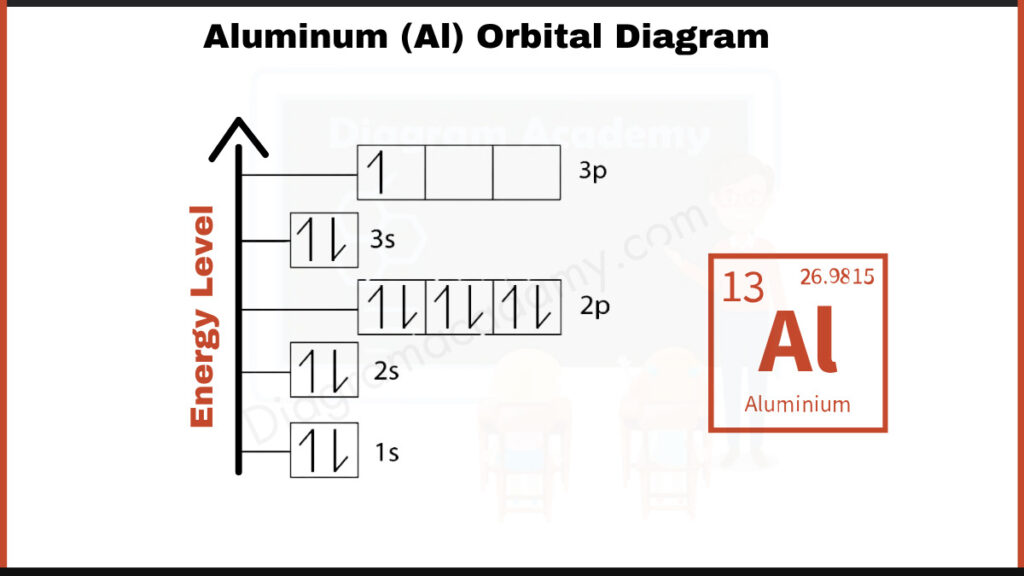
The Full Picture: Aluminum's Electron Configuration
So, the full orbital diagram for aluminum looks like this (simplified explanation): 1s² 2s² 2p⁶ 3s² 3p¹. It’s a shorthand way to show how aluminum’s electrons are arranged.
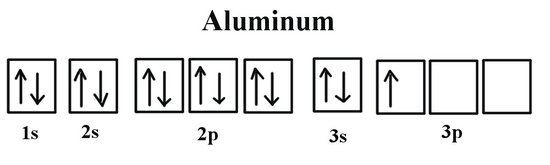
The "superscripts" tell us how many electrons are in each orbital. For example, "2p⁶" means there are six electrons in the 2p orbitals.
That single electron in the 3p orbital is what makes aluminum so special. It's that outermost electron that participates in chemical bonding, determining how aluminum interacts with other elements.
Why Should We Care?
Why bother with all this electron stuff? Because understanding orbital diagrams helps us understand the properties of elements. The way electrons are arranged dictates how an element behaves.
Think about it: that soda can, your phone, even the foil in your kitchen – all thanks to how aluminum shares (or doesn’t share) its electrons! It influences everything from conductivity to reactivity.
Aluminum's lightweight strength makes it ideal for airplanes. This is all thanks to those tiny electrons buzzing around.
Beyond Aluminum: The Electron Neighborhood
The same principles apply to all elements! Each element has its own unique orbital diagram, its own electron arrangement. It’s like each element has its unique personality.
Some elements are generous sharers, others are hoarders. This is what makes chemistry so fascinating and diverse. It's all about those electrons!
By understanding orbital diagrams, we can predict how elements will interact. This helps us create new materials, develop new technologies, and understand the world around us.
Making Chemistry Fun: Visualizing the Atom
Don't be intimidated by the technical terms! Think of orbital diagrams as visual stories about where electrons hang out. It's like a seating chart for the tiniest party imaginable.
There are many interactive tools and diagrams available online that can help you visualize these concepts. Check them out!
Who knew that something so small could be so interesting? Now, the next time you see an aluminum can, you'll know a little more about what's going on inside at an atomic level. Perhaps you'll even whisper a thank you to that lone electron in the 3p orbital for doing its job!
A Final Thought: It's All Connected
The world is made of atoms, and atoms are made of electrons, protons, and neutrons. Understanding these fundamental building blocks helps us understand everything else.
So, embrace the world of chemistry, and don't be afraid to explore the fascinating world of electrons! You might just surprise yourself with what you discover.
From soda cans to spaceships, the orbital diagram of aluminum is a silent testament to the power of the tiny. And with a little imagination, it's pretty fun to explore.
