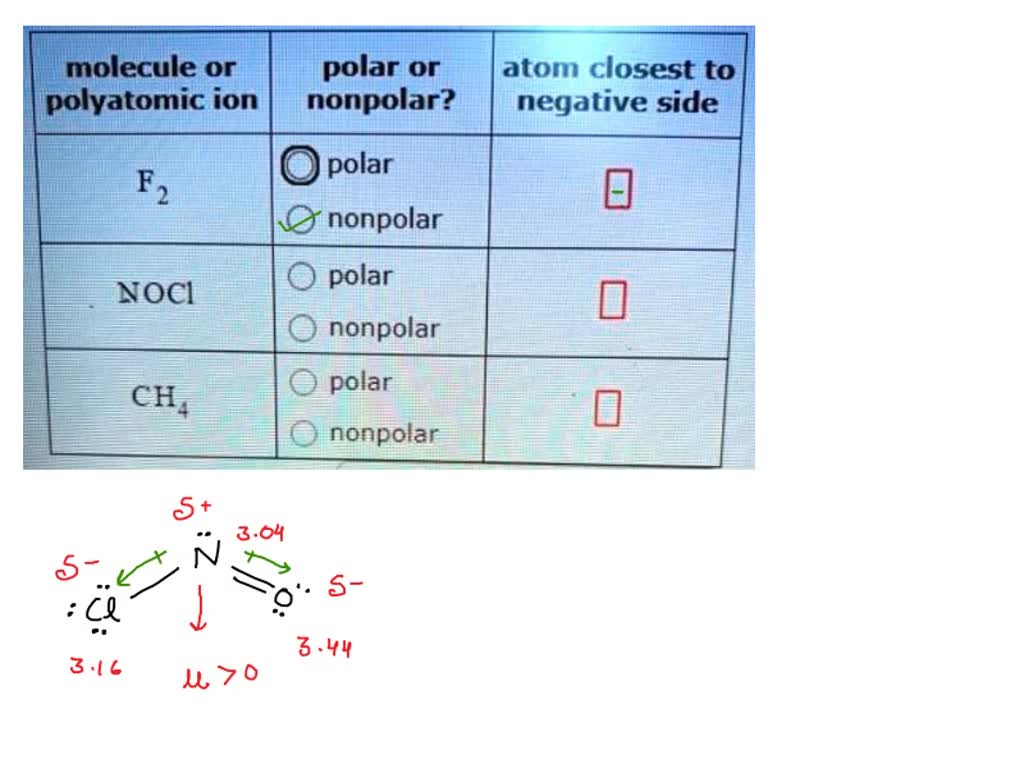
Ocs Atom Closest To Negative Side
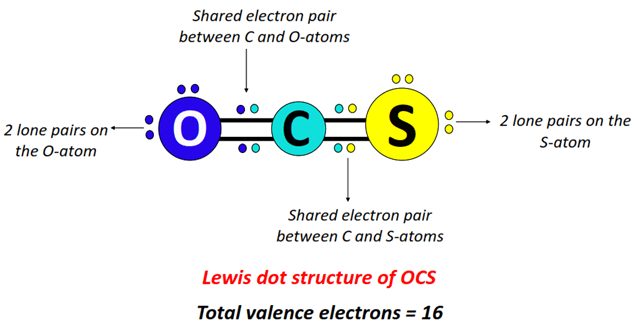
Okay, so pull up a chair, grab a virtual coffee, and let's talk atoms. I know, I know, it sounds like high school chemistry class is back to haunt you. But trust me, this is more like a quantum physics stand-up routine. We're diving into the wild world of elements, specifically the champion of clinginess: Oxygen! Or as I like to call it, "Oxy, the Electron Magnet."
Now, you might be thinking, "Atoms are all neutral, right? Like Switzerland, but smaller?" And you'd mostly be right! Atoms generally have the same number of positively charged protons and negatively charged electrons, balancing each other out. Think of it like a tiny, well-behaved budget. But Oxygen? Oxygen's got... let's just say it has a spending problem when it comes to electrons.
The Charge Game: Why Oxygen's a Little…Negative
Must Read
Here's the lowdown. Atoms crave a full outer shell of electrons. It's like having a complete set of Pokemon cards, or finally finding matching socks in the dryer. This full outer shell makes them stable and happy. Oxygen has six electrons in its outer shell. It needs two more to achieve that sweet, sweet atomic bliss.
This desperation leads to some interesting behavior. Oxygen will practically stalk other atoms, trying to snatch up those precious two electrons. It's like that friend who always "borrows" your charger and never returns it. Except in this case, it's on a fundamental, atomic level! Think of it like atomic kleptomania. "I need those electrons! My preciousss!"

When Oxygen manages to snag those electrons, it becomes negatively charged. Hence, "Oxy, the Electron Magnet." Officially, we say it becomes an "ion" with a negative charge. But really, it's just a slightly grumpy atom that finally got what it wanted (electrons!). This makes it electronegative. Basically, a fancy word for being an electron hog.
Electronegativity: The Scale of Clinginess
Now, let’s put this clinginess into context. Scientists use a scale called "electronegativity" to measure how strongly an atom attracts electrons in a chemical bond. This scale usually goes from 0 to 4. And guess who's chilling near the top? Our pal, Oxygen! It has a whopping electronegativity value of around 3.44. Not the absolute highest (Fluorine steals that crown with 3.98, the show-off!), but still pretty darn high.
Think of it like this: if atoms were contestants on a dating show, electronegativity would be their "level of neediness." Oxygen would be the one writing dramatic poetry and planning the wedding after the first date. Maybe that's a slight exaggeration. (Or is it? The heart, or rather, the atom, wants what it wants!).
So, What’s the Big Deal? Why Should I Care About Oxygen's Electron Obsession?

Well, for starters, without Oxygen's electron-grabbing tendencies, you wouldn't be able to breathe. Seriously! The oxygen you inhale is crucial for a process called cellular respiration, which is how your body generates energy. It's basically atomic-level fuel burning, and Oxygen is the spark that gets the whole combustion going. This whole process relies on Oxygen's strong pull on electrons.
And that's not all! Oxygen's electronegativity is also responsible for: the formation of water (H2O! Remember that stuff?); the way proteins fold in your body (critical for their function!); and, you know, rust. Yes, even rust is thanks to Oxygen's insatiable appetite for electrons. It's like the ultimate frenemy: essential for life, but also slowly corroding your car. Talk about a complicated relationship!
Fun Facts to Impress Your Friends (or Annoy Them)
- Did you know that Oxygen makes up about 21% of the Earth's atmosphere? The remaining is mostly Nitrogen, which is pretty chill about electrons (relatively speaking).
- Oxygen is also the most abundant element in the Earth's crust. It's literally everywhere, constantly trying to bond with something. Like that one overly friendly person at a party.
- Liquid oxygen is blue! I know, right? Mind. Blown.
So, next time you take a deep breath, remember "Oxy, the Electron Magnet," the slightly negative atom that makes life as we know it possible. It might be a bit clingy, but hey, we all have our quirks! And without its electron-grabbing abilities, we'd all be in a lot of trouble. Thank you, Oxygen! For being slightly negative, but ultimately, incredibly important.
And that's the story of Oxygen's slightly negative side. Now, who wants another virtual coffee?
