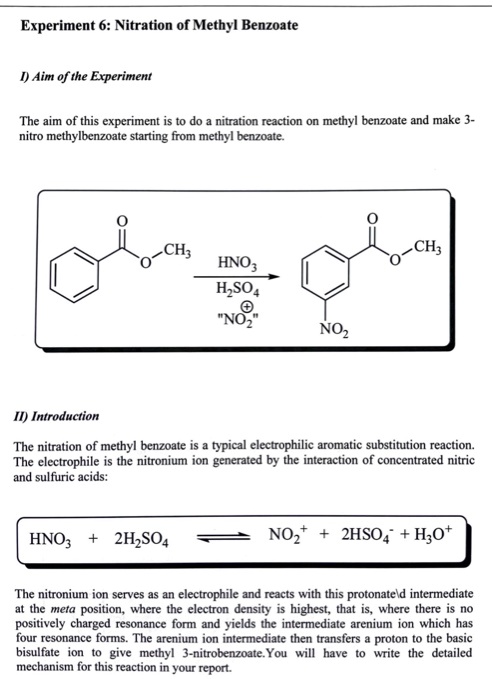
Nitration Of Methyl Benzoate

Ever wondered how scientists create the building blocks for things like perfumes, dyes, and even some life-saving drugs? Well, buckle up, because we're diving into a fascinating chemical reaction called the Nitration of Methyl Benzoate! Sounds intimidating, right? Don't worry, we'll break it down in a way that's easier than baking a cake (and probably just as satisfying!).
Think of it like this: imagine you have a Lego castle (that's our Methyl Benzoate) and you want to add a cool new tower with a flag (that's the Nitro group we're adding). The nitration reaction is the process of carefully attaching that tower to the existing castle structure.
So, what's the big deal? Why do scientists bother doing this? The purpose of nitrating Methyl Benzoate is to add a nitro group (NO2) to the benzene ring. This little addition dramatically changes the properties of the molecule. It's like giving our Lego castle some superpowers!
Must Read
But what are these "superpowers," exactly? Well, nitrated compounds are incredibly versatile. This reaction is a key step in synthesizing a wide range of important products. For example:
- Pharmaceuticals: Many drugs rely on nitrated compounds as key intermediates. They're used in everything from antibiotics to painkillers. Think of it as adding a "healing potion" ingredient to our castle!
- Dyes: The nitro group can significantly alter the color of a molecule, making it perfect for creating vibrant dyes for clothing, inks, and more. Our Lego castle suddenly gets a vibrant new paint job!
- Explosives: Okay, maybe not the most cheerful application, but it's true! Some nitrated compounds are explosive. While we won't be making any dynamite in our kitchen (please don't!), it highlights the powerful effect of the nitro group.
The benefits of performing this reaction stem from its ability to create these essential building blocks. It allows chemists to synthesize complex molecules with specific properties, paving the way for innovation in various fields. The nitration of methyl benzoate is a fundamental skill for organic chemists. This reaction demonstrates the principles of electrophilic aromatic substitution and is often used as a teaching tool in undergraduate laboratories. Students learn important laboratory techniques such as reflux, filtration, and recrystallization. Think of it as learning the secrets to building even bigger and better Lego castles!
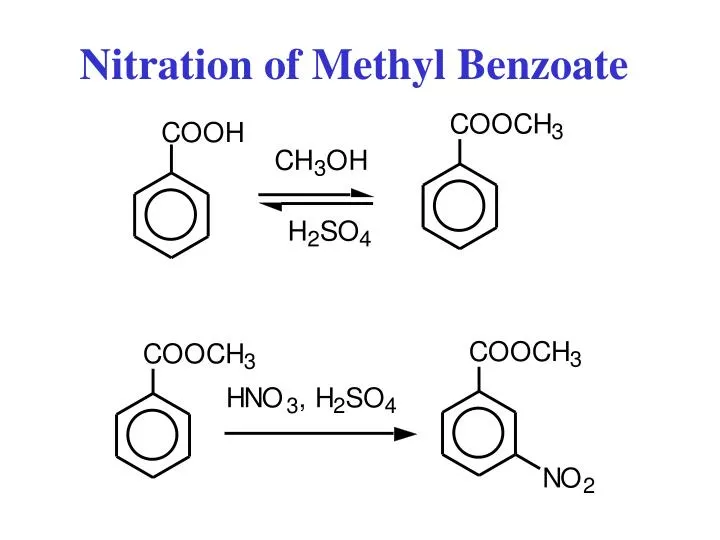
While the actual lab procedure involves carefully mixing Methyl Benzoate with concentrated acids (think sulfuric and nitric acid – handled with extreme care!), the underlying principle is surprisingly simple: we're strategically adding a Nitro group to the benzene ring to create a new and useful molecule. The location of the nitro group on the ring is influenced by the directing effects of the ester group already present on the ring.
So, the next time you encounter a vibrant dye, a life-saving drug, or even just a really cool-looking chemical structure, remember the nitration of Methyl Benzoate – the unsung hero of molecular transformation! It's a testament to the power of chemistry to build, transform, and improve the world around us.