Naome Strong Or Weak Nucleophile

Alright, let's talk about something that might sound intimidating: nucleophiles. But trust me, it's way cooler than it sounds. Think of it like this: in the world of chemistry, we have different personalities, just like in real life. Some are bold and assertive, ready to jump into action, while others are more… reserved. That's pretty much what we're dealing with when we talk about strong versus weak nucleophiles.
So, what exactly is a nucleophile? The name gives it away, right? It means "nucleus-loving." These are molecules or ions with a serious craving for positive charges. They're electron-rich, meaning they've got extra electrons they’re itching to share (or, more accurately, donate). Think of them as the philanthropists of the chemical world! They see a positive charge and they're like, "Here, let me help you out with some electrons!"
Why Should We Care About Strength?
Okay, so we know what nucleophiles are. But why does it matter whether they're strong or weak? Well, imagine you’re trying to organize a party. A strong nucleophile is like that super enthusiastic friend who's already making the decorations and inviting everyone before you've even decided on a theme. A weak nucleophile? They're more like, "Yeah, a party sounds nice... eventually."
Must Read
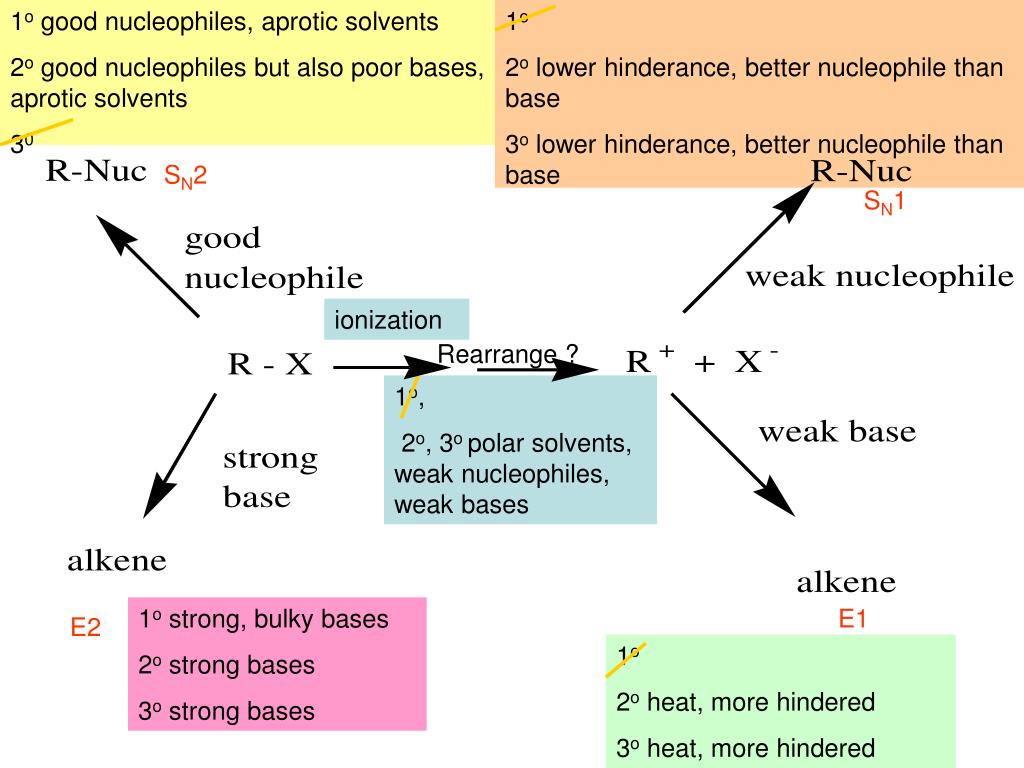
In chemistry, a strong nucleophile will react faster and more readily than a weak one. This affects the whole reaction pathway, what products you get, and how efficient the whole process is. Pretty important, right?
Think of it like ordering food. A strong nucleophile is like ordering online – quick, efficient, and you get what you want relatively fast. A weak nucleophile is like calling in an order to a busy restaurant on a Friday night. You might get your food eventually, but it’s going to take a while, and there’s a chance they’ll mess it up!

What Makes a Nucleophile Strong?
So, what gives a nucleophile its strength? A few key things come into play:
- Charge: This one's pretty straightforward. A negatively charged nucleophile is generally stronger than a neutral one. Why? Because it's already got that extra electron density and is just itching to use it. Think of it as having a head start in a race!
- Electronegativity: Electronegativity is how strongly an atom attracts electrons to itself. If the atom bearing the negative charge is highly electronegative (like oxygen in water), it holds onto those electrons more tightly. This makes it a weaker nucleophile. Why share when you like hoarding? A less electronegative atom (like carbon) is more willing to donate.
- Steric Hindrance: This fancy term just means how bulky the molecule is. A really bulky nucleophile might have trouble getting close enough to the positive charge to react. It’s like trying to squeeze a giant beach ball through a narrow doorway – not gonna happen easily! Smaller, less hindered nucleophiles are usually stronger.
- Solvent Effects: The solvent (the liquid the reaction is happening in) can also play a role. Some solvents can stabilize nucleophiles, making them less reactive. It's like putting your energetic friend in a calming sensory deprivation tank - they're not going to be as enthusiastic about the party anymore!
Sodium: Friend or Foe? (Well, Neither, Actually)
You mentioned Sodium (Na). Now, sodium itself isn't a nucleophile. It's a metal that readily loses an electron to become a positive ion (Na+). However, sodium is often used in compounds that create strong nucleophiles. For example, sodium hydroxide (NaOH) in solution generates hydroxide ions (OH-), which is a strong nucleophile.
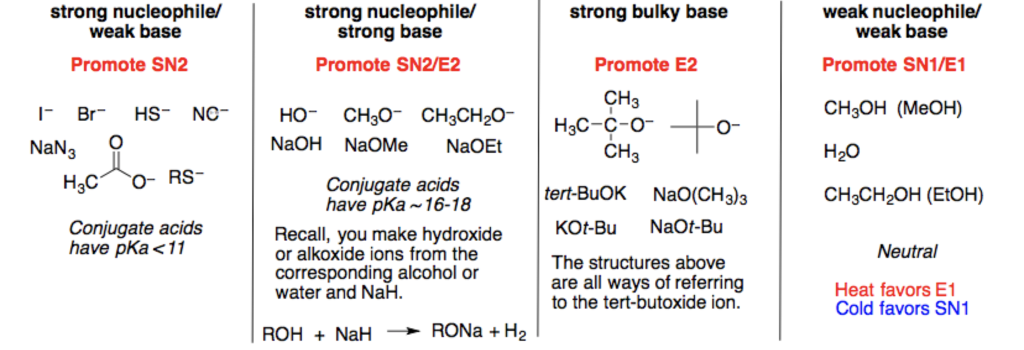
Think of sodium like the matchmaker. It helps bring the nucleophilic part (like OH-) into existence and makes it ready to go! So, while sodium isn't directly the nucleophile, it's often a key player in the game.
So, Why is This Cool?
Why is understanding nucleophiles and their strength important, or even cool? Because it lets us control chemical reactions! By carefully choosing our nucleophiles and solvents, we can direct reactions to go the way we want them to. We can synthesize new drugs, create new materials, and understand how biological processes work. It's like being a conductor of a chemical orchestra, using nucleophiles as your instruments to create beautiful (and useful) music!
Isn't it amazing how understanding something as seemingly abstract as nucleophilic strength can have such a huge impact on the world around us? From the simplest reactions in a test tube to the most complex processes in our bodies, these little electron donors are playing a crucial role. So next time you hear the word "nucleophile," don't run away! Embrace the electron-loving power and remember: it's all about that positive charge!