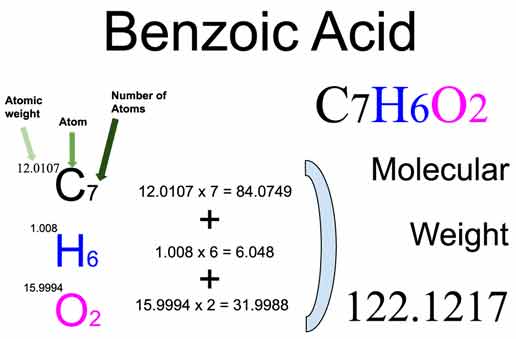
Molecular Weight Benzoic Acid

Ever wondered what goes on behind the scenes to keep your food fresh or ensure your beauty products last? Chemistry, of course! And at the heart of many of these processes lies a humble molecule, a real workhorse in the world of chemistry: benzoic acid. Learning about it, and even its molecular weight, isn't just for scientists in lab coats. It's a fascinating peek into the invisible world that affects our daily lives.
So, what is benzoic acid and why should we care? Simply put, it's a naturally occurring organic acid found in many plants and animals. You might even be surprised to know that it's in some of the fruits you eat! But its real value comes from its ability to inhibit the growth of bacteria, yeast, and mold. This makes it an exceptional preservative.
The benefits are clear: longer-lasting food and products! Think about it. Have you ever noticed "sodium benzoate" listed on the ingredients of your favorite soda, juice, or even pickle jar? That's a salt derived from benzoic acid, doing its job to keep those goodies fresh and safe to consume. Beyond food, benzoic acid is also utilized in cosmetics and personal care products. It helps prevent the growth of microorganisms that could spoil the product or, worse, harm your skin. It is also used to manufacture other chemicals.
Must Read
Now, let's talk about that "molecular weight" we mentioned earlier. The molecular weight of benzoic acid is approximately 122.12 grams per mole (g/mol). This number is crucial for chemists. Imagine you're trying to replicate a scientific experiment that uses benzoic acid. Knowing its molecular weight allows you to accurately measure the correct amount needed for the reaction to work. It's a fundamental concept in chemistry, underpinning calculations for everything from synthesizing new drugs to understanding chemical reactions.
Where might you encounter benzoic acid in an educational setting? It's a common subject in introductory chemistry courses. Students might use it in experiments to learn about titration, acid-base chemistry, or organic synthesis. Its relatively simple structure makes it a good starting point for understanding more complex organic molecules. Plus, it's a good example of how theoretical knowledge translates into practical applications.

Want to explore this fascinating molecule further? Here are a few simple tips:
- Read ingredient labels: Scan the ingredient list of common foods and personal care products. See how often you can spot "benzoic acid" or "sodium benzoate."
- Look up its structure online: Search for images of the benzoic acid molecule. Visualizing its shape can help you understand its properties.
- Investigate scientific experiments: There are many simple experiments using benzoic acid that you can find online, often suitable for high school or introductory college chemistry.
While you might not become a chemist overnight, understanding the basics of benzoic acid and its molecular weight is a fun and accessible way to connect with the world of chemistry that's all around us. It's a reminder that science isn't just something that happens in labs; it's an integral part of our daily lives.