Molecular Orbital Diagram For Be2

Okay, picture this: you're at a party, right? And you see this REALLY shy person in the corner. Like, glued-to-the-wall shy. You think, "Hey, maybe they just need someone to talk to!" So you go over and try to spark a conversation. But…nothing. Zilch. They just kinda…exist. That, my friends, is the molecular embodiment of Be2. Sort of. Stick with me here.
What I’m getting at is sometimes things look like they should work, but they just…don't. And that's where understanding molecular orbital (MO) diagrams comes in handy. Especially when you're trying to figure out why some molecules are stable and others are, well, as exciting as watching paint dry. (No offense to paint-drying enthusiasts out there.)
So, What’s the Deal with Be2?
Be2, or diberyllium, is a fascinating little molecule. On paper, it should exist. Beryllium, after all, has two valence electrons. So two beryllium atoms should be able to form a bond, right? Think of it like two shy wallflowers finally deciding to dance! But reality isn't always that straightforward, is it? That’s because of those MO diagrams.
Must Read
Molecular orbital diagrams are like blueprints for understanding how atoms combine to form molecules. They show us the energy levels of the molecular orbitals that are created when atomic orbitals interact. Remember atomic orbitals? Those s, p, d, and f orbitals? Yeah, those guys. (Don't worry if you're a bit rusty; just think of them as regions of space where electrons like to hang out.)
Building the Be2 MO Diagram
Let's break down the Be2 MO diagram step by step. Beryllium has the electron configuration 1s22s2. Now, we're mostly concerned with the valence electrons, which are the 2s electrons in this case. The 1s electrons are core electrons and don't participate in bonding.

When two beryllium atoms come together, their 2s atomic orbitals combine to form two molecular orbitals: a bonding σ2s orbital and an antibonding σ2s* orbital. The bonding orbital is lower in energy than the atomic orbitals, meaning it's more stable. The antibonding orbital is higher in energy, making it less stable.
Think of it like this: the bonding orbital is like a comfy couch where the electrons want to relax. The antibonding orbital is like a wobbly chair that no one wants to sit on. Guess where the electrons are going to go?
Filling in the Electrons
Each beryllium atom contributes two electrons, so we have a total of four electrons to place in our molecular orbitals. We follow the same rules as filling atomic orbitals: fill the lowest energy levels first (Aufbau principle) and only put two electrons in each orbital (Pauli exclusion principle).

So, we fill the σ2s bonding orbital with two electrons, and then we fill the σ2s* antibonding orbital with the remaining two electrons. (Are you still with me? This is the crucial part!)
The Bond Order Tells the Story
Now comes the magic: the bond order. The bond order tells us how many bonds are formed between the two atoms. It's calculated as:

Bond Order = (Number of electrons in bonding orbitals - Number of electrons in antibonding orbitals) / 2
In the case of Be2, we have two electrons in the bonding orbital and two electrons in the antibonding orbital. So:
Bond Order = (2 - 2) / 2 = 0
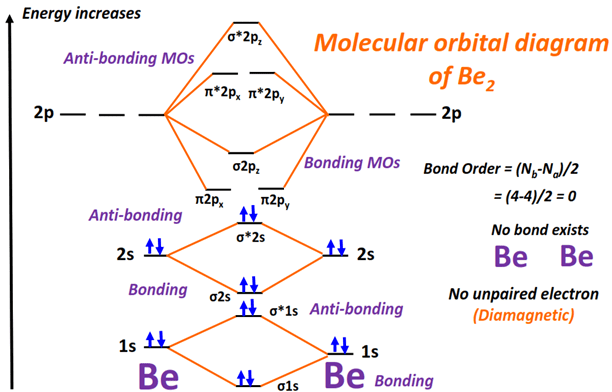
A bond order of zero means… no bond! Zip. Nada. This is why Be2 is so incredibly unstable. It's energetically unfavorable for the beryllium atoms to bond together. They're happier existing as individual atoms. Like those shy wallflowers deciding they’d rather be alone.
Why Bother Learning This?
Understanding MO diagrams helps us predict the stability and properties of molecules. Sure, Be2 might seem like a weird, almost-non-existent exception, but the principles we learn from it apply to countless other molecules. From the oxygen we breathe to the plastics in our phones, MO theory helps us understand how chemical bonds work. And isn't that just a little bit mind-blowing?
So, next time you're feeling a little… unreactive, just remember Be2. Sometimes, things are just better off staying single. And that's perfectly okay. Now, go forth and bond with some knowledge! (And maybe someone at the party, if you're feeling up to it.)
