Molecular Mass Of Cuso4 5h2o

Ever stared at a mysterious ingredient in a recipe or maybe a label on a cleaning product and felt a little lost? Chemistry can seem like a whole different language sometimes! But don't worry, we're going to unravel a tiny piece of that language today: the molecular mass of CuSO4·5H2O. Yeah, that looks like a mouthful, but trust me, it's less intimidating than it sounds. Think of it as unlocking a secret code to understand what things really are!
So, What is CuSO4·5H2O Anyway?
Okay, let's break it down. CuSO4 is copper sulfate. Ever seen those vibrant blue crystals used in root killers or sometimes in science kits? That’s often copper sulfate. And the "·5H2O" part? That just means that for every one molecule of copper sulfate, there are five molecules of water attached to it. Think of it like a group of friends all sticking together – copper sulfate and its five water buddies!
This whole combo is officially called copper(II) sulfate pentahydrate. Don't worry about memorizing that! Just remember it's copper sulfate with some water hanging around. The water is actually bound to the copper sulfate in a specific way, making it part of the overall structure. It's not just wet copper sulfate; it's a compound with the water as an integral part.
Must Read
Why Should I Care About Its Molecular Mass?
Good question! Why bother with this at all? Well, understanding molecular mass is fundamental to understanding how much of something you need for a reaction, a solution, or even just to follow instructions correctly. Think of baking: if you add too much flour or not enough sugar, your cake won't turn out right. Molecular mass is like the baking measurements for chemistry. It tells you the "weight" of one mole (a specific number) of the molecule.
Imagine you're making a plant food solution using copper sulfate pentahydrate to help prevent fungal diseases. Knowing the molecular mass helps you calculate exactly how much of the stuff to dissolve in the water to get the correct concentration. Too little, and it won't work. Too much, and you could harm your plants. Getting it right is key, and that's where molecular mass comes in!

Calculating the Molecular Mass: It's Easier Than You Think!
Now, for the nitty-gritty: calculating the molecular mass. This is where we add up the atomic masses of all the atoms in the compound. Don't panic! We're going to take it step-by-step, and you can always use a periodic table for reference.
Here's what we need to know:
- Copper (Cu): Atomic mass ≈ 63.55 atomic mass units (amu)
- Sulfur (S): Atomic mass ≈ 32.07 amu
- Oxygen (O): Atomic mass ≈ 16.00 amu
- Hydrogen (H): Atomic mass ≈ 1.01 amu
Okay, so let’s break down CuSO4·5H2O:
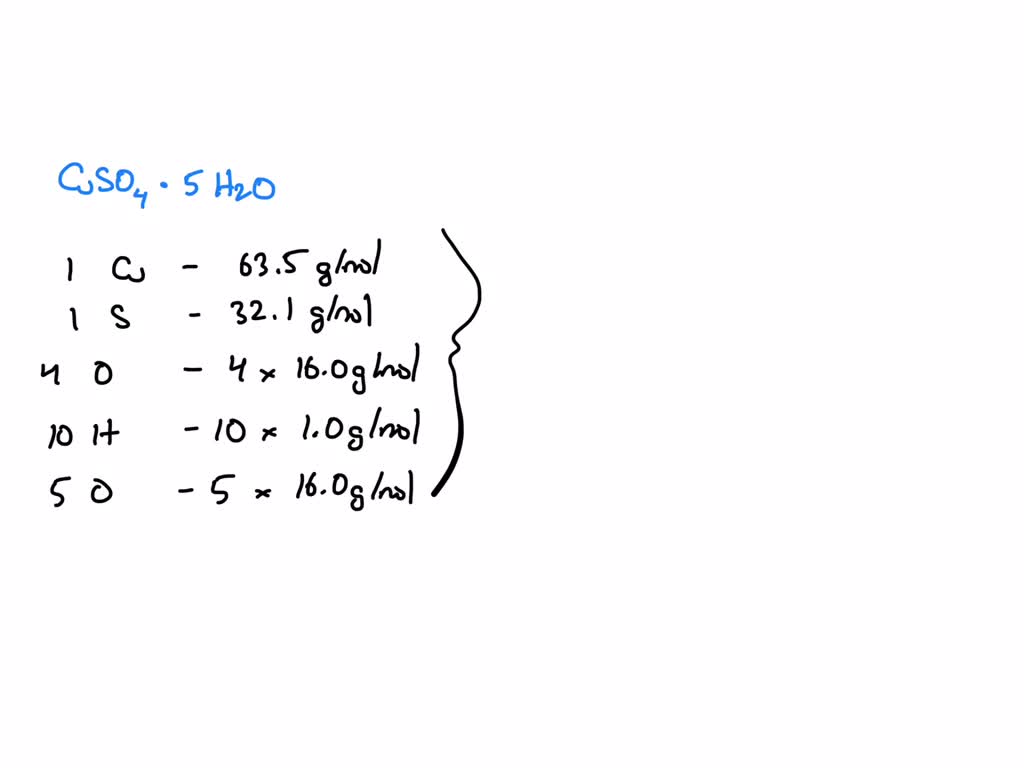
- 1 Copper (Cu): 1 x 63.55 amu = 63.55 amu
- 1 Sulfur (S): 1 x 32.07 amu = 32.07 amu
- 4 Oxygen (O) from CuSO4: 4 x 16.00 amu = 64.00 amu
- 5 Water (H2O) molecules: Each water molecule has 2 Hydrogen (2 x 1.01 amu = 2.02 amu) and 1 Oxygen (1 x 16.00 amu = 16.00 amu). So, one H2O is 18.02 amu. Therefore, 5 H2O is 5 x 18.02 amu = 90.10 amu
Now, add it all up: 63.55 + 32.07 + 64.00 + 90.10 = 249.72 amu
So, the molecular mass of CuSO4·5H2O is approximately 249.72 amu. If you were using grams instead of amu, you'd be calculating the molar mass, which is 249.72 grams per mole (g/mol). Same number, just a different unit for a bigger quantity!

Real-World Applications and Why It Matters
Beyond plant food solutions, copper sulfate pentahydrate has a bunch of uses. It's used in algicides to control algae growth in ponds and pools. It's sometimes used in animal feed as a supplement. It's even used in some medical applications, although that's less common.
Understanding its molecular mass is crucial for accurately formulating these products. Too much copper sulfate can be toxic, while too little might be ineffective. So, even if you're not a chemist, knowing a little about molecular mass can help you appreciate the science behind the products you use every day.
Think of it this way: next time you see something involving chemistry, whether it's in your garden, your kitchen, or even a science fiction movie, you'll have a little bit more understanding of what's going on behind the scenes. You'll know that molecular mass is a key concept that helps scientists (and even everyday folks) measure and understand the world around them. And that's pretty cool, right?

