Molecular Geometry Of H2s
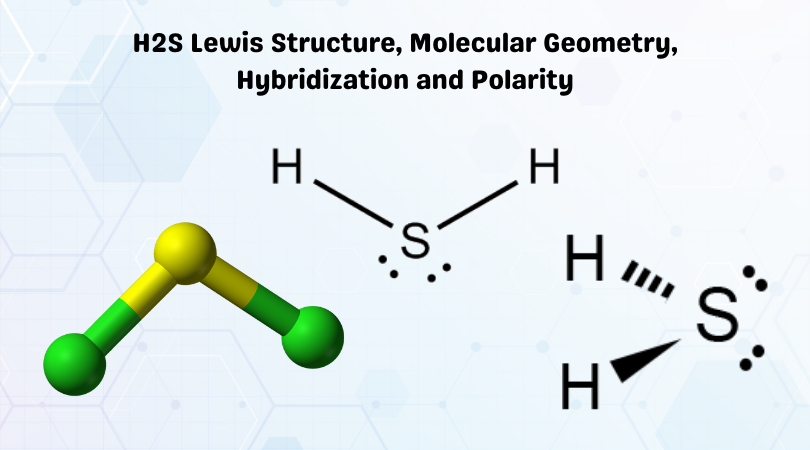
Alright, let's talk about Hydrogen Sulfide (H2S). Yeah, that rotten egg smell. But stick with me, it gets interesting...kinda.
H2S: The Bent Bandit
We’re diving into its molecular geometry. Sounds intimidating, right? Don't worry, it's basically just how the atoms are arranged. Think of it like arranging your furniture, but with less back pain.
Everyone says H2S is bent. BENT. Like a poorly executed yoga pose. This is because of those pesky lone pairs on the sulfur.
Must Read
The Lone Pair Conspiracy
So, Sulfur has these extra electrons, chilling out in pairs. These lone pairs are like super-clingy friends. They take up more space and repel the bonding pairs (the ones holding hands with Hydrogen) pushing them closer together.
This "bent" shape is the accepted wisdom. But I have a confession: I’m not entirely convinced it's that bent.
Hear me out!
My Unpopular Opinion: H2S Isn't THAT Bent!
Now, I know, I know. Textbooks scream "bent!" Professors dramatically gesture a bend with their hands. Everyone nods in agreement.
But think about it. It's not like H2S is doing the limbo under a limbo stick. It's more of a slight lean, a gentle curve.

Sure, those lone pairs are there, pushing things around. But are they really exerting that much influence?
Is It Really That Different From Water (H2O)?
Everyone compares it to water (H2O). Water, also bent, is the golden child of molecular geometry. Teachers coo over its polarity.
H2O gets all the attention. But is the difference between the bond angle in H2O and H2S that extreme?
I propose the bend is exaggerated! Maybe it's more of a subtle curve. A gentle arc. A sophisticated slouch.
The Case for "Slightly Less Bent"
Consider the poor hydrogens! They're just trying to bond nicely. Maybe they're putting up a fight against those domineering lone pairs.

Perhaps, just perhaps, they're managing to straighten things out a little more than everyone gives them credit for.
Maybe, in the grand scheme of molecular shapes, H2S is just misunderstood. It's not a blatant bender; it's simply...slightly less straight.
Let's Redefine "Bent"
I think it's time for a new category in molecular geometry. Something between "linear" and "bent". Something like "slightly askew."
Or maybe "gently persuaded from linearity." H2S could be the poster child!
Think of it like this: Is a slightly crooked picture on the wall still a crooked picture? Or is it just… slightly off?

The Rotten Egg Revelation
Now, I'm not saying H2S is suddenly going to become linear. The lone pairs are still there, doing their thing.
And yes, it will still smell like rotten eggs. My hypothesis about its 'less bent' nature won’t change that, sadly.
But maybe, just maybe, we can all agree that the "bentness" of H2S might be a little overblown. A smidge exaggerated. A tad dramatized.
Join the Revolution!
Who's with me? Let's start a movement. #H2SIsNotThatBent.
We can write strongly worded letters to textbook publishers. We can stage a protest outside chemistry departments. (Just kidding… mostly.)

Okay, maybe not. But the next time you think about H2S, remember this: it might not be as bent as you think.
In Conclusion: A Matter of Perspective
Molecular geometry is all about perspective, right? One person's "bent" is another person's "graceful curve."
So, embrace the slightly-less-bent nature of H2S. Appreciate its subtle deviations from linearity.
And remember, even rotten egg smells can have a surprisingly interesting backstory.
So next time you smell that distinctive aroma, you'll know it's not just H2S, it's H2S: the misunderstood, the slightly-less-bent, the molecule that dares to defy expectations. You're welcome.
