Molecular Geometry Of Brf3

Okay, picture this: You're building with LEGOs, right? You've got all these different shapes, and you're trying to make something awesome. But if you just slap them together randomly, it's gonna be a total mess. You need to think about how they connect, how the shapes influence each other, and how the whole thing balances. That's kind of like molecules! Except instead of LEGO bricks, we're talking about atoms, and instead of trying to build a spaceship, we're figuring out their molecular geometry.
And speaking of interesting shapes... let's talk about Bromine Trifluoride, or BrF3. Ever heard of it? Probably not. But trust me, its shape is way cooler than a simple pyramid. It's more like a... well, we'll get to that.
Why Does Shape Even Matter?
Good question! (Glad you asked!) The shape of a molecule is super important because it dictates how it interacts with other molecules. Think about it – a key only fits into a specific lock because of its shape. Similarly, the shape of BrF3 determines how it reacts with other chemicals, its physical properties (like melting point), and pretty much everything else it does. So, understanding its geometry unlocks a whole new level of understanding about its behavior.
Must Read
Enter VSEPR Theory: The Shape Whisperer
So, how do we know what shape BrF3 is? We use something called VSEPR theory, which stands for Valence Shell Electron Pair Repulsion theory. Basically, it says that electron pairs (both bonding pairs and lone pairs) around a central atom will try to get as far away from each other as possible. Why? Because they're all negatively charged, and like charges repel! It's like having a bunch of toddlers; they want their personal space.
Remember that LEGO analogy? This is where it comes in. Just like the LEGO pieces need to find a stable configuration, the electron pairs around the Bromine atom (Br) need to arrange themselves to minimize repulsion.
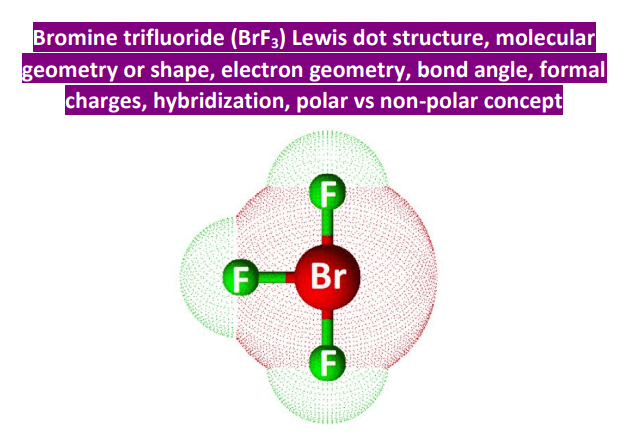
BrF3: A Molecular T-Rex (Sort Of)
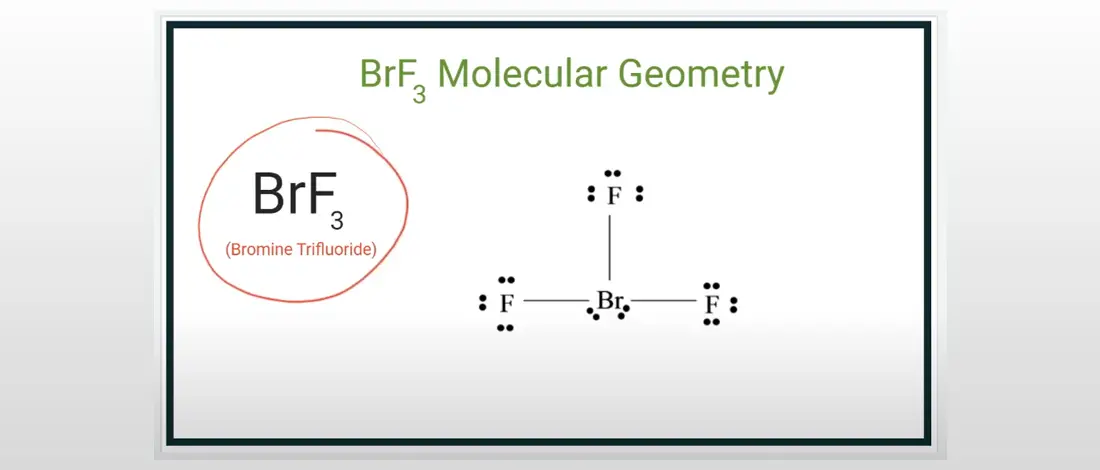
Now, let's apply VSEPR theory to BrF3. Bromine (Br) is the central atom. It has 7 valence electrons. Three of them bond with Fluorine (F) atoms (forming 3 bonding pairs). That leaves us with…four electrons, or two lone pairs. Boom! Big deal! (Spoiler: those lone pairs will seriously mess with our expectations)
If we only had bonding pairs, BrF3 might be a trigonal planar molecule (like BF3). But those lone pairs? They take up more space than bonding pairs because they're only attached to one atom (Br), so they have more freedom to spread out and repel. Those little troublemakers!

So, what happens? The lone pairs push the bonding pairs closer together, resulting in a T-shaped geometry. Yes, a T-shape! Imagine the Bromine in the middle of the T, with Fluorine atoms at the bottom and at the ends of the horizontal line. The lone pairs are lurking in the equatorial plane (sort of above and below the Br). It's officially a T-Shaped Molecule.
Seriously, isn't that kind of cool? From what seemed like it should be, to the actual, wonky, T-shape it really becomes? Nature is crazy.
Why is This Important?
BrF3 is a powerful fluorinating agent. Knowing its shape helps us understand why it's so reactive. The T-shape might contribute to how it attacks other molecules. Moreover, understanding VSEPR theory and applying it to BrF3 helps us predict the shapes of other molecules, and therefore, their properties and reactivity.
So, the next time you're playing with LEGOs (or just staring blankly at the ceiling), remember BrF3 and its bizarre T-shape. It's a reminder that even something as seemingly simple as a molecule can have a surprising and interesting structure, and that understanding that structure unlocks a world of chemical possibilities.
Who knew chemistry could be so... oddly shaped? 😉

