Molecular Geometry Of Bcl3
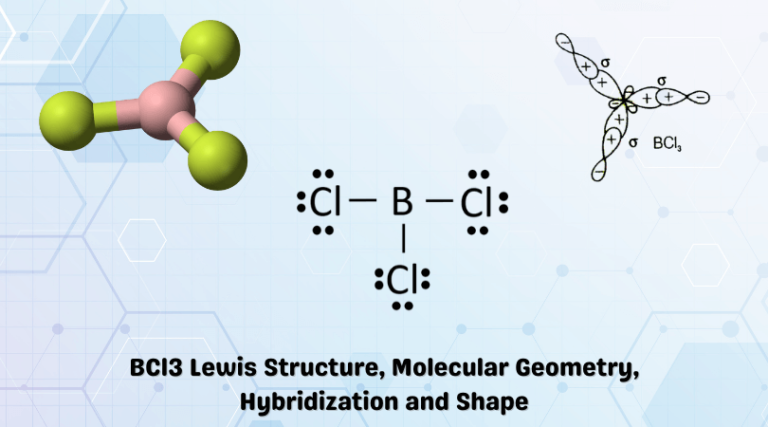
Let's talk about BCl3, or Boron Trichloride, if you're feeling fancy. It's a molecule. And molecules have shapes! Think of it like LEGOs, but with electrons and invisible forces.
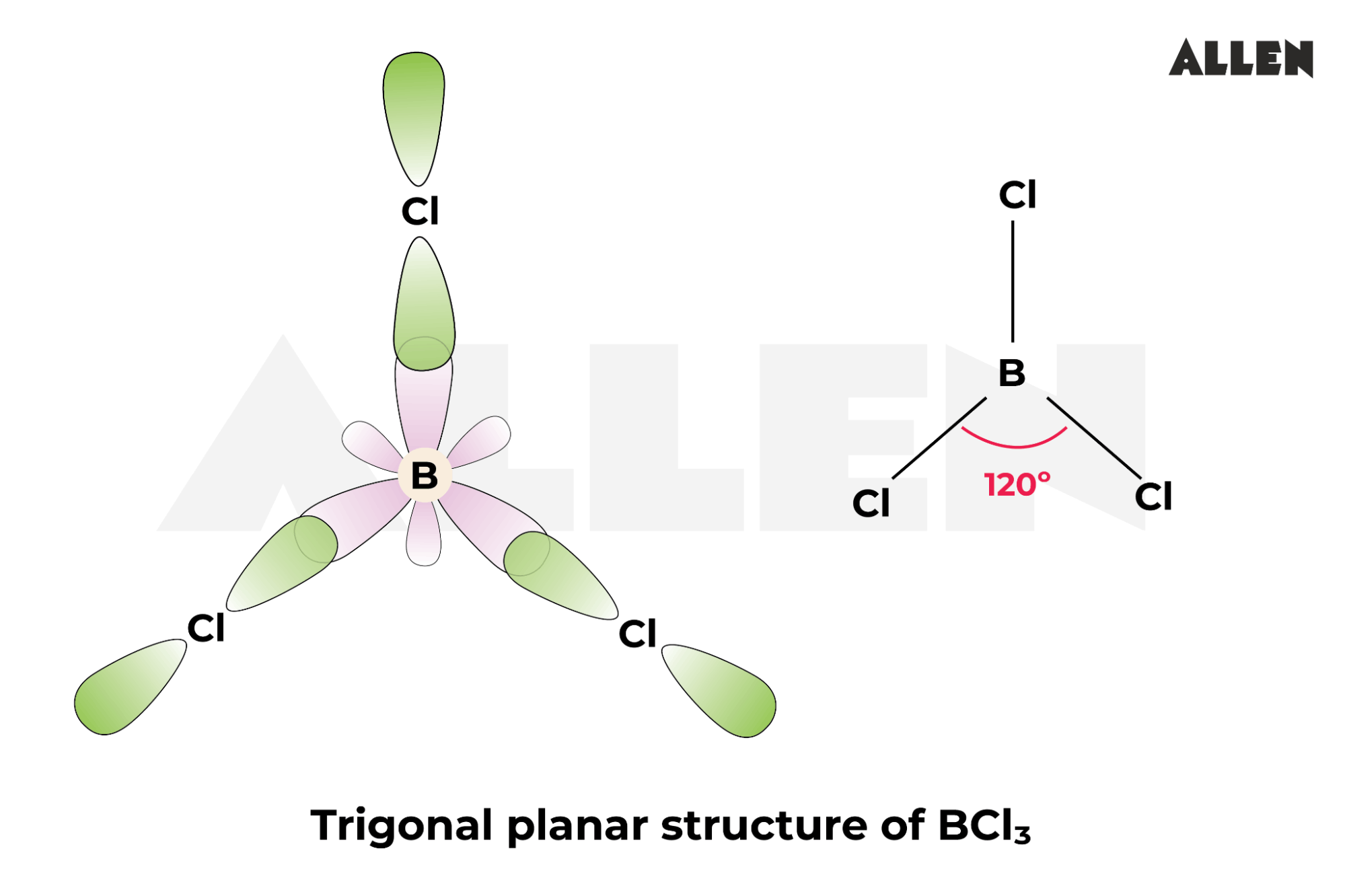
Most folks describe BCl3 as having a trigonal planar shape. Textbook perfect! All angles exactly 120 degrees. So… symmetrical and boring? Maybe.
My Hot Take: BCl3 is Slightly…Lopsided?
Okay, hear me out. I know the textbooks disagree. But do textbooks ever do the dishes? I suspect not. So, I'm taking their opinion with a grain of (sodium chloride) salt.
Must Read
The classic description says Boron sits perfectly in the middle. With three Chlorines equally spaced around it. A flawless, flat triangle of atomic bliss. I remain unconvinced.
Electronegativity: The Culprit?
We know that Chlorine is a bit of an electron hog. A greedy little atom, pulling electrons towards itself. Boron, bless its heart, is a bit more…chill. It's happy to share (well, mostly).
So, each Chlorine is tugging on Boron's electrons. That pull isn't perfectly even, is it? It's a three-way tug-of-war. Maybe not exactly identical on each side. Think of it like three toddlers trying to steal the same cookie. It's chaotic!

My (unpopular) opinion? Those Chlorines are distorting the molecule! They are not created equal. Some may be closer to the boron than others!
Lone Pairs: The Red Herring?
Ah, the absence of lone pairs! The classic argument for BCl3’s perfect symmetry. Boron has no extra electrons hanging around, messing things up.
But that doesn't mean the shared electrons are perfectly balanced. The Chlorines are still doing their electron-thieving thing. Their electron cloud is more negatively charged than the Boron.

It’s like saying a perfectly balanced seesaw is unaffected by a strong wind. The wind will affect it, just like electronegativity will affect the electron cloud!
Evidence? I Rest My Case…Sort Of
Look, I'm not a quantum physicist. I barely passed high school chemistry. But intuition is worth something, right?
Think about real-world molecules, vibrating, jiggling, and bumping into each other. At that scale, is anything truly perfect? It's highly doubtful, in my opinion.
Could there be slight variations in bond length and angles? Absolutely! And I will keep arguing that point until they put me in chemistry jail! Okay, maybe not that far. But I feel pretty sure about it.
But Wait, There’s More!
Consider the reactions of BCl3. It loves to grab extra electrons and form adducts. Wouldn't a slight imbalance in the molecule make it more reactive?
A perfectly symmetrical molecule might be less inclined to react. A little bit of distortion could create a more attractive spot for incoming electrons. Just a theory!
Plus, nobody ever talks about the isotopes! Are all Boron atoms exactly the same? Or Chlorine atoms? Slight variations can matter.

Conclusion: Embrace Imperfection!
So, there you have it. My radical theory about the slightly lopsided nature of BCl3. Don’t quote me on this at any professional setting. Seriously, don’t.
The textbooks say trigonal planar. Fine. But I think there is a lot going on behind the scenes that nobody talks about. Things are more complicated.
Maybe, just maybe, the universe is a little less perfect than we think. And that's okay! It’s what makes chemistry, and life, interesting. So, join me. Embrace the imperfect! Let’s create a revolution of slightly-lopsided-molecule lovers!
"The best things in life are a little bit messed up." -Probably someone who studies chemistry (or should!)
