Molar Mass Of Potassium Hydrogen Phthalate

Okay, okay, I know what you're thinking: "Molar mass? Potassium hydrogen phthalate? Sounds like a snoozefest!" But trust me, understanding this stuff is like unlocking a secret level in the world of... well, okay, maybe not gaming. But definitely in baking, gardening, and even crafting! It's all about getting precise measurements, and that's where the fun (and accuracy) begins.
So, what's the big deal about molar mass and potassium hydrogen phthalate (KHP for short)? Simply put, molar mass is the weight of one mole (a specific number – think of it as a chemist's dozen) of a substance. KHP is a really handy acid that's used to figure out the exact concentration of other solutions. We use its molar mass to know precisely how much KHP we're using, which then tells us exactly how much of the other substance we're dealing with.
For beginners, understanding molar mass is a foundational step in chemistry. It's like learning your ABCs before writing a novel. Knowing how to calculate the molar mass of KHP (or anything else!) means you can confidently perform calculations in experiments and understand the relationships between different chemicals. Think of it as unlocking a cheat code for your science homework!
Must Read
Now, how does this apply to families or hobbyists? Imagine you're making soap. Precise measurements are crucial for a safe and effective product. While you might not be using KHP directly, understanding the concept of molar mass will help you accurately measure ingredients and avoid potentially harmful reactions. Or, perhaps you're into hydroponics. Calculating the correct amount of nutrients to add to your water is essential for healthy plant growth. Again, the principles of molar mass are at play!
Let's talk about variations. KHP is just one example. You can calculate the molar mass of any compound! Water (H₂O), table salt (NaCl), even sugar (C₁₂H₂₂O₁₁). The process is the same: identify the elements, find their atomic masses on the periodic table, and multiply by the number of atoms of each element in the formula. Then, add it all up!
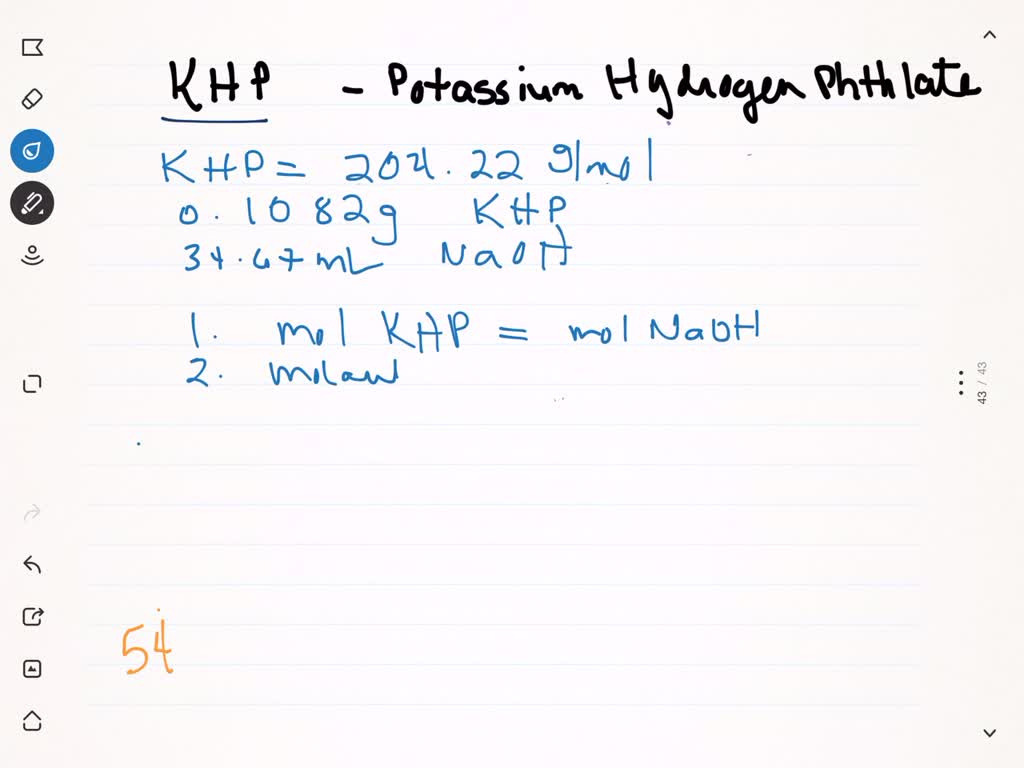
Here are some simple, practical tips to get started: First, get a periodic table! It's your best friend. Second, practice! Start with simple compounds like water and gradually work your way up to more complex ones. Third, double-check your work. A small mistake in a calculation can throw everything off. Finally, use a calculator! Nobody wants to do all that multiplication by hand.
So, while "molar mass of potassium hydrogen phthalate" might not sound like the most thrilling topic at first, understanding its relevance opens up a world of possibilities. From ensuring accurate measurements in your DIY projects to grasping fundamental concepts in chemistry, the knowledge you gain is surprisingly valuable. Plus, there's a certain satisfaction in knowing you can confidently calculate something so precise. It's like having a superpower...a very nerdy, but undeniably useful, superpower!



