Molar Mass Of Naphthalene
Hey there, science adventurers! Ever feel like chemistry is this mystical, complicated beast? I get it. But trust me, there are some really cool, fundamental concepts that, once you grasp them, make the whole world of molecules a lot less intimidating (and even, dare I say, fun!). Today, we're diving into one of those key concepts: molar mass. And to make it even more exciting, we're going to explore it using a super interesting molecule – naphthalene!
Naphthalene? What's that, you ask? Well, you might not know it by name, but you definitely know its smell. It's the stuff that makes mothballs so… mothbally! Okay, maybe "pleasant" isn't the right word, but it's certainly distinctive. And that distinctive smell comes from a very specific arrangement of carbon and hydrogen atoms, all held together in a nice, stable structure. Think of it like the LEGOs of the molecular world.
So, What's Molar Mass Anyway?
Alright, let’s break it down. Molar mass is simply the mass of one mole of a substance. Now, a mole isn't that cute, burrowing creature you might be picturing (although those are adorable too!). In chemistry, a mole is a specific number – 6.022 x 1023 to be precise. We call it Avogadro's number. It's a gigantic number! It represents the number of atoms, molecules, or ions in one mole. Imagine having that many LEGOs! You could build, well, just about anything.
Must Read
Why do we need such a huge number? Because atoms and molecules are incredibly tiny! We can't weigh them individually on a regular scale. So, the mole gives us a way to relate the mass of a substance we can measure in the lab (in grams) to the number of individual particles (atoms, molecules, etc.) that are present.
Think of it like buying eggs. You don't usually buy individual eggs; you buy them by the dozen. A "dozen" is a convenient unit to group things together. A mole is the same thing, just on a much, much, much grander scale!
Finding Naphthalene's Molar Mass: A Step-by-Step Adventure
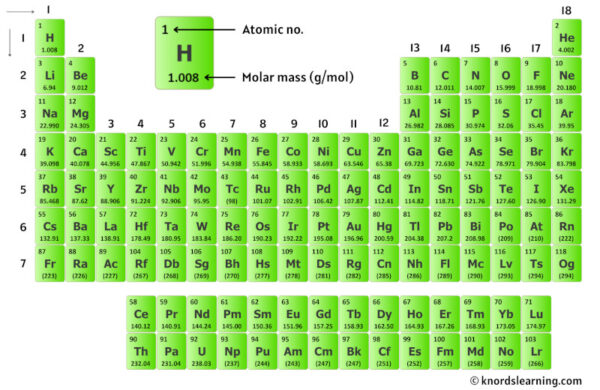
Okay, let's get practical. Naphthalene's chemical formula is C10H8. This tells us that each molecule of naphthalene contains 10 carbon (C) atoms and 8 hydrogen (H) atoms. Got it? Great!

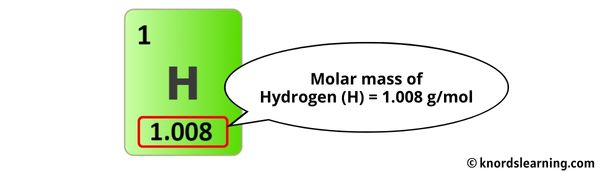
To find the molar mass, we need to look up the atomic masses of carbon and hydrogen on the periodic table. Don't worry, it's not as scary as it sounds! The periodic table is your friend! You'll find that the atomic mass of carbon is approximately 12.01 atomic mass units (amu) and the atomic mass of hydrogen is approximately 1.01 amu.
Now for the fun part – the calculation! To get the molar mass of naphthalene, we do the following:
Molar mass of C10H8 = (10 x atomic mass of C) + (8 x atomic mass of H)

Plugging in the values:
Molar mass of C10H8 = (10 x 12.01 amu) + (8 x 1.01 amu)
Molar mass of C10H8 = 120.1 amu + 8.08 amu
Molar mass of C10H8 = 128.18 amu
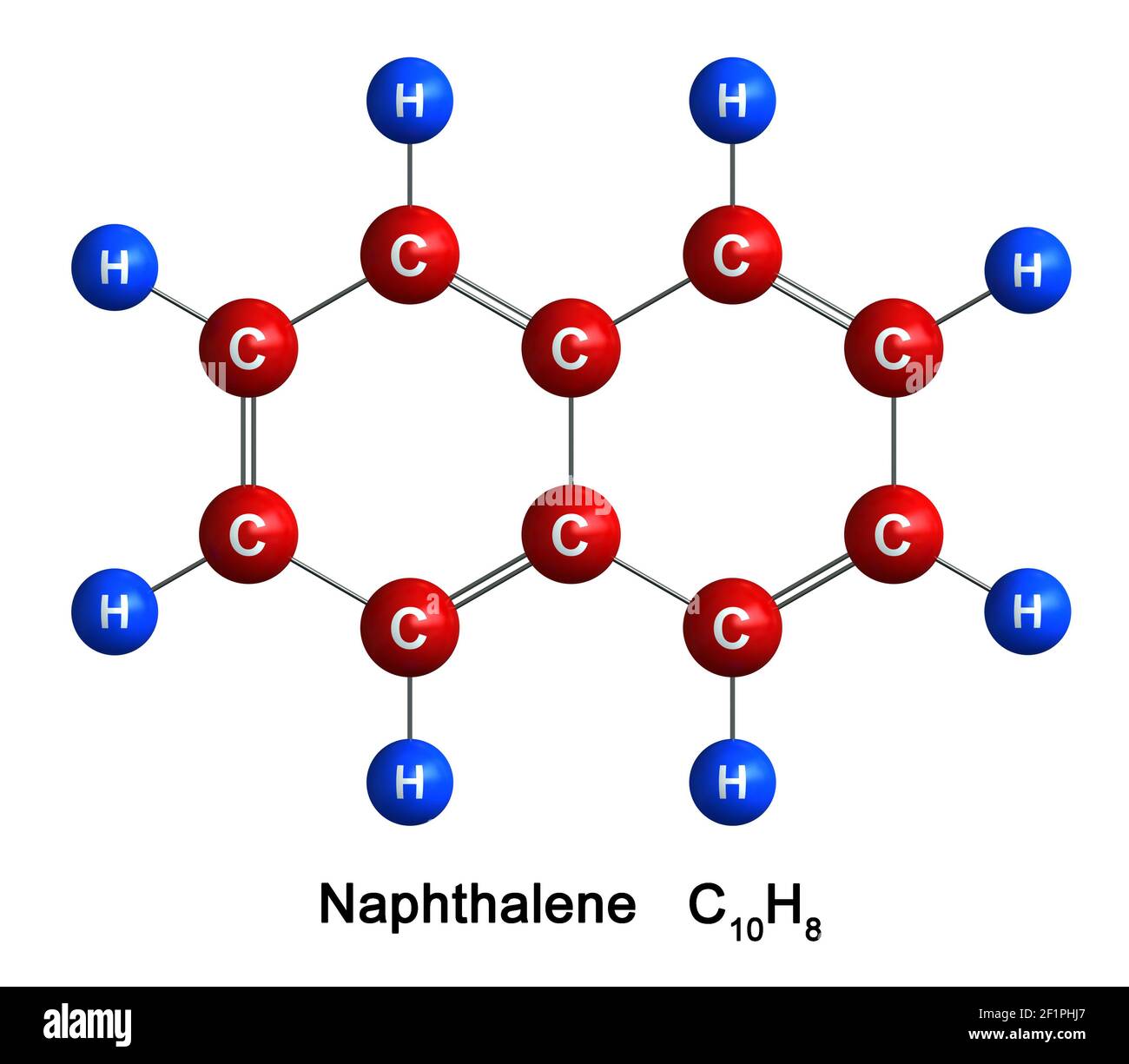
But wait! We want the molar mass in grams per mole (g/mol), not amu. Luckily, the numerical value is the same! One mole of naphthalene has a mass of 128.18 grams. Boom! We did it!
Why Should You Care About Molar Mass?
Okay, you might be thinking, "That's cool and all, but why does this matter in real life?" Well, understanding molar mass is absolutely crucial for so many things! It's the foundation for calculating how much of a substance you need for a chemical reaction, for determining the concentration of a solution, and for understanding the composition of different materials. Basically, it's a core concept in chemistry, and chemistry is everywhere!
Think about cooking. Recipes tell you how much of each ingredient to use. Molar mass is like having the perfect conversion chart for the molecular world. It allows chemists to measure out just the right amount of reactants to get the desired products. No more exploding beakers (hopefully!).
Or consider pharmaceuticals. Knowing the molar mass of a drug is essential for determining the correct dosage. Too little, and it won't be effective. Too much, and it could be harmful. Getting it just right can literally save lives!
See? Suddenly, mothballs and molar mass don't seem so boring after all! It's all connected!
By understanding molar mass, you're unlocking a fundamental tool for understanding the world around you. You're empowering yourself to explore the chemistry of everything from the food you eat to the medicines you take. And that's pretty awesome, right?
So, go forth and embrace the world of moles, molecules, and molar mass! Don't be afraid to ask questions, explore new concepts, and have fun with it. You might just surprise yourself with what you can discover. And who knows, maybe one day you'll be the one making groundbreaking discoveries in chemistry! The possibilities are endless!
