Molar Mass Of Ascorbic Acid

Okay, let's talk about vitamin C. Or, as the science folks like to call it, ascorbic acid. Sounds intimidating, right? But trust me, we can handle this.
Specifically, we're going to sneakily explore its molar mass. Don’t run away screaming! It’s not as scary as your last tax bill.
The Great Ascorbic Acid Adventure
Ascorbic acid, our sunshine vitamin, is made of a few things. Carbon, hydrogen, and oxygen. Like a tiny, delicious superhero team.
Must Read
Now, each of these elements has a specific weight. We find these on the periodic table. That chart your high school teacher threatened you with.
Think of it like this: each element has a price tag. To find the total price of the molecule, you need to know how many of each item are needed.
Unpopular Opinion Time!
Here’s my completely unnecessary, yet totally heartfelt opinion: math is okay. I said it! Don't hate me.
Especially when it involves something as cool as figuring out what something weighs at a molecular level.
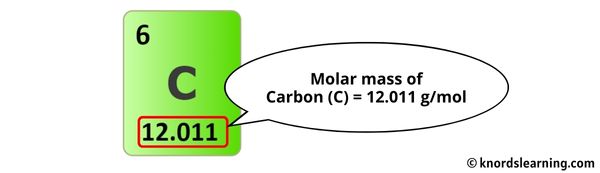
Okay, back to business. Ascorbic acid's formula is C6H8O6. This means six carbons, eight hydrogens, and six oxygen atoms are involved.
The Numbers Game
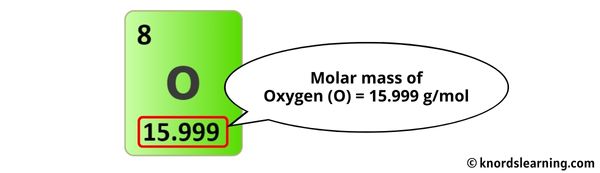
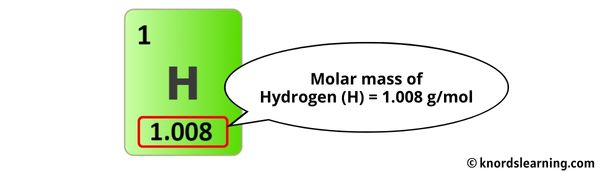
Each carbon (C) has an atomic mass of about 12.01 atomic mass units (amu). Hydrogen (H) is roughly 1.01 amu each. Oxygen (O) is approximately 16.00 amu a pop.
We need to do some multiplying. Six carbons at 12.01 each gives us 72.06. Eight hydrogens at 1.01 each makes 8.08. Six oxygens at 16.00 each gives us a grand total of 96.00.
Add those values all up: 72.06 + 8.08 + 96.00. Are you ready for this? The answer is 176.14 amu. That's the molar mass!
Wait, What's Molar Mass Again?
Good question! The molar mass represents the mass of one mole of a substance.
A mole is just a really, really big number of molecules. Avogadro’s number, if you’re asking, but no pressure to remember that.

So, 176.14 amu is basically saying that one mole of ascorbic acid weighs about 176.14 grams. That's it. We did it!
Why Bother?
Okay, you might be thinking, "Great, I know the molar mass of ascorbic acid. How does this help me order a pizza?" It doesn't. At all.
But knowing this is super important if you are creating things in a lab. Chemists use it to calculate how much of a substance they need for reactions.
Imagine baking a cake but not knowing how much flour to add! Chaos would ensue.
In Conclusion (and More Opinions)
So, next time you’re popping a vitamin C pill, give a little nod to the molar mass of ascorbic acid. It's quietly working hard in the background.
Another unpopular opinion: science is actually quite fun! Especially when you can relate it to everyday things, like your daily dose of vitamin C.
And finally, a very serious declaration: everyone deserves a slice of cake after doing some calculations. It’s practically the law.
"The pursuit of knowledge is a lifelong adventure, so don’t be afraid to explore the world around you." - Some Wise Person, Probably.
Now, go forth and spread the word about the wonders of molar mass! Or, you know, just enjoy your vitamin C.
