Melting Point And Freezing Point Are The Same
Alright, gather 'round, folks! Let me tell you about something that blew my mind the other day. It's about melting and freezing. You know, those things your ice cream does when you forget it on the counter, and that water does when you leave it outside in December.
So, I was chatting with my super-smart friend, let's call him Professor Know-It-All (because, well, he kind of is). And he drops this bomb on me: "Melting point and freezing point? They're the same, you know."
I almost choked on my latte. "Wait, what? That can't be right! One's going from solid to liquid, the other's going from liquid to solid. They're, like, total opposites!"
Must Read
He just smirked that infuriatingly knowing smirk. "Nope. Same temperature. It's all about perspective, my friend."
Perspective is Key (and Maybe a Thermometer)
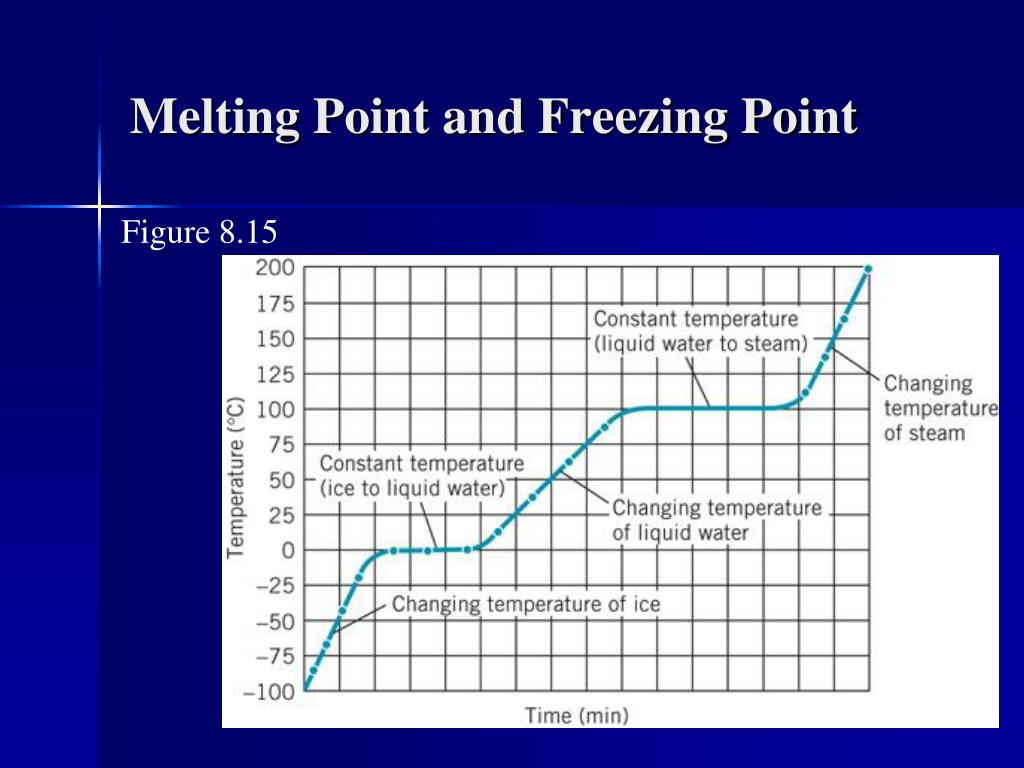
Think about it. Imagine you’re heating up an ice cube. It’s sitting there, minding its own frozen business. As the temperature rises, BAM! It hits 0 degrees Celsius (or 32 degrees Fahrenheit for my American friends who still haven't embraced the metric system… just kidding! Mostly).
What happens? The ice starts to melt, right? That 0°C is its melting point. The temperature stays there while it’s changing to water.
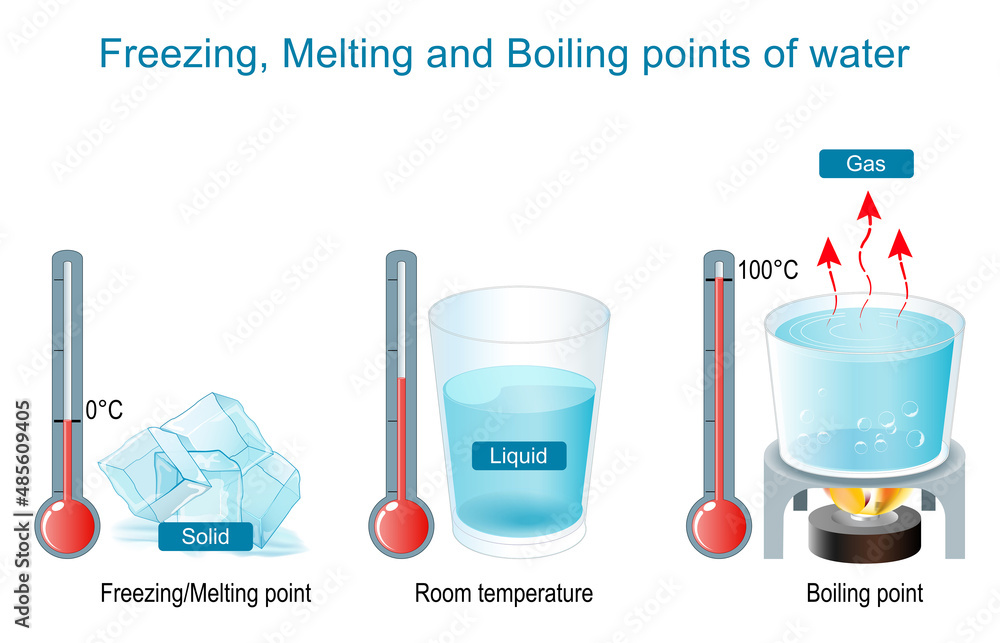
Now, imagine you’re cooling down some water. It's happily sloshing around at room temperature. But as you chill it, something magical (or rather, scientific) happens. Again, you hit 0°C. And… the water starts to freeze! Boom! Freezing point! The temperature stays there as it is changing to ice.
See? Same temperature! It's like deciding whether to call the glass half-empty or half-full. It’s the same glass, just seen from a different angle.
But What About... Everything Else?
Okay, okay, I know what you're thinking. What about all those other substances? Gold? Iron? That weird goo that sticks to your shoe sometimes?

Well, the principle is the same. Every substance has its own unique melting/freezing point. Gold melts at a scorching 1,064°C (1,947°F). That goo on your shoe? I don’t even want to know, but I bet it’s nasty and probably has a low melting point!
The important thing to remember is that the temperature at which a substance changes from solid to liquid is the exact same temperature at which it changes from liquid to solid.
Exceptions to the Rule (Because Science Likes to Be Complicated)
Now, just when you think you've got it all figured out, science throws you a curveball. Because, well, that's what science does.

There are some substances that can be a little finicky. Factors like pressure and impurities can slightly affect the melting/freezing point. For example, if you're way up on a mountain where the air pressure is lower, water will boil at a lower temperature. And adding salt to ice lowers its freezing point, which is why we do it on icy roads.
Think of it like this: the melting/freezing point is like a stubborn toddler. It usually does what it's supposed to, but sometimes it throws a tantrum and needs a little coaxing (in the form of pressure or impurities).
The Takeaway: Impress Your Friends at Parties!
So, the next time you're at a party and the conversation lulls (because let's face it, that always happens), you can casually drop this knowledge bomb. "Did you know that the melting point and freezing point are the same?"

Watch as your friends’ jaws drop in amazement. You'll be the life of the party! Or, at least, the person who knows a random science fact.
And if someone challenges you, just tell them to ask Professor Know-It-All. Or, you know, just Google it. But where's the fun in that?
Seriously though, understanding melting and freezing points is pretty cool (pun intended!). It helps us understand how the world works, from making ice cream to forging metal. So go forth and spread the knowledge! And maybe grab an ice cream while you're at it.
Just don't forget it on the counter, or you'll have to explain the melting point all over again.
