Magnesium Phosphate Molar Mass

Ever wondered about the secret lives of molecules? Buckle up! We're diving into the surprisingly captivating world of magnesium phosphate molar mass. Yes, you read that right. It's more thrilling than you think!
Okay, okay. Molar mass sounds a bit…sciency. But trust me. It’s like a VIP pass to understanding how much stuff is actually in a tiny speck of magnesium phosphate. Think of it as the molecule's weight. It’s given in grams per mole (g/mol). A mole is a chemist's way of saying "a whole bunch" of molecules. It's about 6.022 x 1023, to be exact. That's a lot of particles!
What Makes Magnesium Phosphate So Special?
Magnesium phosphate isn't just some random chemical compound. It's a team player in biology and medicine. It can exist in various forms, each with slightly different formulas and molar masses. This variety is part of what makes it so interesting.
Must Read
Think of it like this: You have a basic recipe for a cake. Magnesium is like flour, phosphate is like sugar. But, you can add different types of flour (different forms of magnesium) and different types of sugar (different forms of phosphate) to create slightly different cakes (different forms of magnesium phosphate).
One common form is trimagnesium phosphate, with the formula Mg3(PO4)2. Now, let's get to the exciting part: figuring out its molar mass. Ready for some number crunching (the fun kind, I promise)?
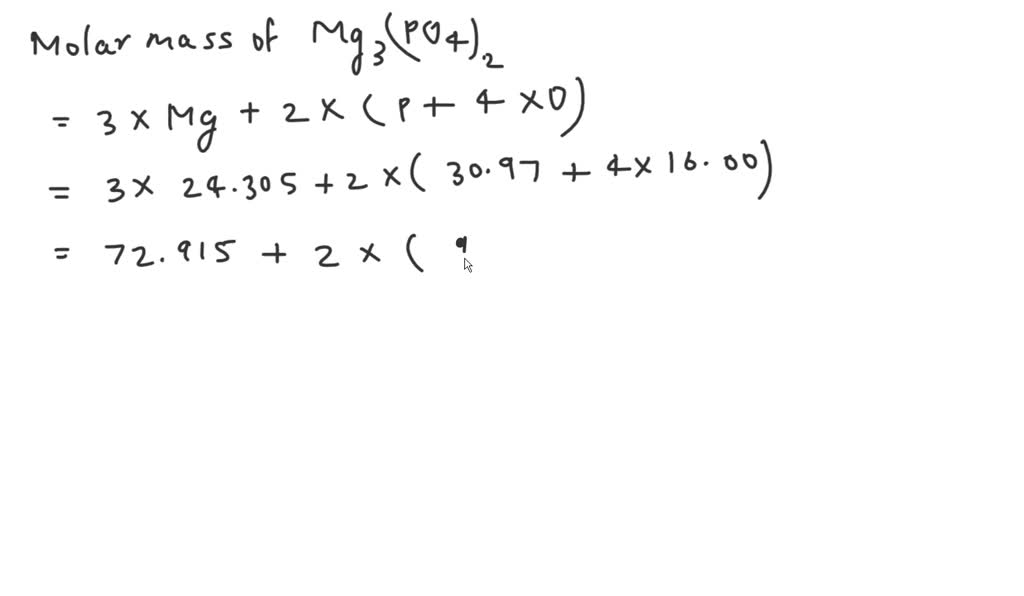
To calculate the molar mass, you'll need a periodic table. It's your map to the atomic weights of each element in the compound. The atomic weight is like the weight of a single atom of that element.
Here's the breakdown:

- Magnesium (Mg): Atomic weight is about 24.31 g/mol. Since there are three magnesium atoms, that's 3 * 24.31 g/mol = 72.93 g/mol
- Phosphorus (P): Atomic weight is about 30.97 g/mol. There are two phosphorus atoms, so that's 2 * 30.97 g/mol = 61.94 g/mol.
- Oxygen (O): Atomic weight is about 16.00 g/mol. There are eight oxygen atoms (2 x 4), which gives us 8 * 16.00 g/mol = 128.00 g/mol.
Now, add them all up: 72.93 + 61.94 + 128.00 = 262.87 g/mol. Ta-da! The molar mass of trimagnesium phosphate is approximately 262.87 g/mol. Pretty neat, right?
Why Bother with Molar Mass?
“Okay, great," you might be thinking. "But why should I care?” Well, imagine you're a scientist conducting an experiment. You need to know exactly how much magnesium phosphate to use to get the right results. Molar mass is your guiding light. It allows you to convert between mass (what you can weigh on a scale) and the number of molecules (what actually reacts).
Think of baking! If you want to make a cake that tastes just right, you need to measure the ingredients accurately. Molar mass is like the recipe's ingredient list for chemical reactions. It tells you the correct proportions of each element to use. Without it, your experiment could be a flop! (Or, in baking terms, a burnt cake).
![Magnesium phosphate [Mg3(PO4)2] Molar Mass](https://knordslearning.com/wp-content/uploads/2022/08/1-768x499.jpg)
Different applications require different forms. And each form has a specific molar mass. Consider magnesium hydrogen phosphate. It's a different beast altogether, and its molar mass will be different because it has a different chemical formula.
Molar mass isn't just some abstract concept. It's a fundamental tool used by scientists, engineers, and even some chefs (molecular gastronomy, anyone?) to understand and manipulate the world around us.

The Takeaway
So, the next time you hear the phrase "molar mass," don't run for the hills. Embrace the awesomeness! It's a crucial piece of the puzzle for understanding the chemical world. It's the key to unlocking the secrets of how much "stuff" is in a given sample of a compound. And it all starts with a simple calculation.
Dive deeper. Explore the different types of magnesium phosphate and their molar masses. You might just find yourself hooked on the surprisingly captivating world of molecular weights! Go ahead, be a molecule detective! The world of chemistry awaits!
“The important thing is to never stop questioning." - Albert Einstein
So, go forth and calculate! You might be surprised at what you discover.
