Low Temperature Synthesis Using Molten Salt

Imagine you're a kid again, making mud pies. You mix dirt (your raw materials), water (your solvent), and maybe some glitter (because, why not?) to create something… earthy. Now, imagine that water is actually melted salt, heated just right. Sounds a bit… salty, right?
That, in essence, is the surprisingly accessible world of low-temperature molten salt synthesis. Forget the giant, super-hot furnaces you might picture. We’re talking temperatures lower than what it takes to bake a pizza!
Melting Away the Complexity
Okay, so maybe the “mud pie” analogy is a slight oversimplification. But the core idea holds true. We’re using melted salts as a special kind of liquid environment – a solvent, if you will – to help different ingredients react and form new materials.
Must Read
Think of it like a dating app for atoms. The molten salt brings them together in a cozy, warm environment, making it easier for them to connect and form bonds. Without the molten salt, they might just ignore each other, too busy with their own thing.
And the best part? Because we're working at lower temperatures, we can make materials that would be impossible to create in traditional, super-hot ovens. It's like discovering a whole new set of LEGO bricks that only work at a specific, lower temperature.
The Salty Secret Sauce
What makes these molten salts so special? Well, for one, they're incredibly versatile. There's a whole range of different salts you can use, each with its own unique properties. It’s like having a spice rack for chemistry!
Some salts are better at dissolving certain materials, while others are better at promoting specific types of reactions. It’s all about finding the right salt (or salt mixture) to create the perfect conditions for your desired material to form.

And because the temperature is relatively low, we can use simpler, cheaper equipment. Forget the expensive, high-tech furnaces! We can often get away with something that resembles a glorified hot plate. Talk about budget-friendly science!
Unexpected Benefits: A Happy Accident?
Sometimes, the most amazing discoveries happen by accident. And the world of molten salt synthesis is no stranger to happy coincidences. Imagine a researcher trying to make one material, and instead, they accidentally stumble upon something even better!
Because the process is so sensitive to the specific conditions, even slight variations in temperature or salt composition can lead to unexpected results. It’s like baking a cake and accidentally creating a whole new type of pastry!
This element of surprise is one of the things that makes this field so exciting. You never know what you might discover next! It’s like being a kid again, playing in the dirt and stumbling upon a hidden treasure.

Cleaning Up Our Act: Sustainability is Key
Beyond the scientific marvel, there’s a real push towards making this process more sustainable. Traditional methods of material synthesis often involve harsh chemicals and generate a lot of waste.
Molten salt synthesis, on the other hand, can be much cleaner and greener. We can often recycle the salts and reuse them, minimizing waste and reducing our environmental impact. It's like finding a way to compost our chemical byproducts!
And because we're using lower temperatures, we also save energy. It's like switching from incandescent light bulbs to LEDs – a small change that can make a big difference. This is one area of science with the potential to be better for all of us!
From Batteries to Beyond: The Future is Salty
So, what are we actually using these molten salt-made materials for? The possibilities are endless! One of the most promising areas is in battery technology.

Imagine batteries that can charge faster, last longer, and are safer to use. Molten salt synthesis is helping us create new electrode materials that could revolutionize the way we power our devices and vehicles.
But that's just the tip of the iceberg. These materials are also being explored for use in solar cells, catalysts, and even medical implants. It’s like discovering a magic ingredient that can be used to solve a whole range of problems.
A Salty Tale of Collaboration
The field of low-temperature synthesis is a truly collaborative effort. Scientists from all over the world are working together to push the boundaries of what's possible.
Researchers are sharing their knowledge and expertise, building upon each other's discoveries, and constantly innovating. It’s like a global potluck dinner, where everyone brings a dish to share and everyone benefits from the delicious feast.
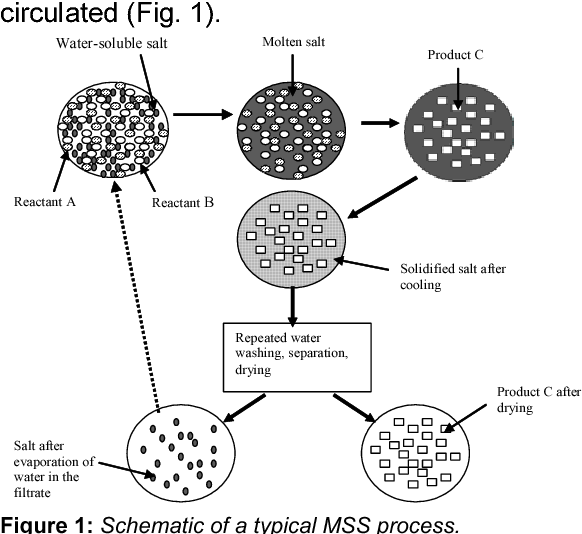
This spirit of collaboration is essential for tackling some of the biggest challenges facing our world today, from climate change to sustainable energy. It is a reminder that the best science happens when we work together.
Embrace the Salty Side
The next time you're reaching for the salt shaker, take a moment to appreciate the amazing potential of this humble mineral. It's not just for seasoning your food; it's also a key ingredient in a revolutionary approach to material synthesis.
From unexpected discoveries to sustainable practices, molten salt chemistry is offering a fresh, and sometimes salty, perspective on how we create the materials that shape our world. It's a reminder that even the simplest ingredients can hold the key to unlocking incredible possibilities.
So, embrace the salty side of science and prepare to be amazed by the transformative power of melted salts. It may just change the world, one atom at a time.
