Lewis Structure Of Ccl2f2
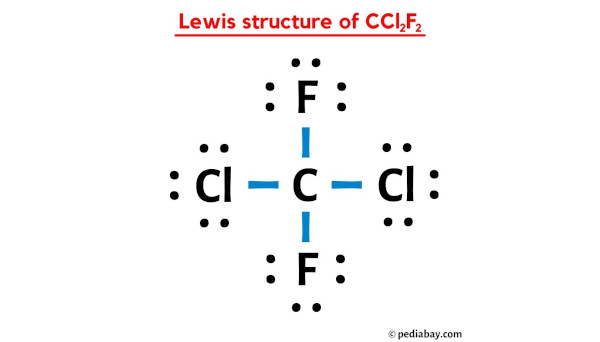
Okay, folks, let's talk about something that might sound a little intimidating – the Lewis structure of CCl2F2. Don't run away! It's not as scary as it sounds. Think of it as a recipe for a molecule, a visual guide to how all the atoms in CCl2F2 are connected and sharing ingredients (electrons!).
What's CCl2F2 Anyway?
CCl2F2 is short for dichlorodifluoromethane. Sounds like a mouthful, right? You might know it better as Freon-12, a once-popular refrigerant. Now, before you start thinking about complicated chemistry equations, let’s relate this to something we all understand: our refrigerators! For a long time, chemicals like CCl2F2 were the unsung heroes keeping our food cold.
Now, the catch: turns out Freon-12 wasn't so friendly to the Earth's ozone layer. Think of the ozone layer as Earth's sunscreen – it protects us from harmful UV rays. CCl2F2, when released into the atmosphere, would float up and start breaking down the ozone. This is why it's now phased out and replaced with more ozone-friendly alternatives. So, understanding its structure helped us realize its impact and find better solutions. That's why even a peek into molecular structure can be beneficial.
Must Read
Building the Lewis Structure: A Molecular Lego Set
So, how do we draw this "recipe," this Lewis structure? Think of it like building with Lego bricks, but instead of colorful plastic, we're using atoms and electrons. Each atom wants to be happy and stable, and that usually means having a certain number of electrons around it.
Carbon (C) is the central atom in CCl2F2. It's like the main character in our story, needing four connections (think of four Lego studs on top) to be content.
We also have Chlorine (Cl) and Fluorine (F). Both of these are a bit like supporting actors, each wanting just one connection (one Lego brick attaching to them).

Here's the game: we need to arrange these atoms so that everyone is happy and has the right number of connections (electrons) around them. Each connection is a shared pair of electrons, a "covalent bond."
Step-by-Step: Let's Draw It!
1. Count the Valence Electrons: This is like counting your ingredients. Carbon has 4, each Chlorine has 7, and each Fluorine has 7. So, 4 + (2 x 7) + (2 x 7) = 28 valence electrons in total. These are the electrons we have to play with.
2. Place the Central Atom: Put Carbon (C) in the middle. It's the star of our show!
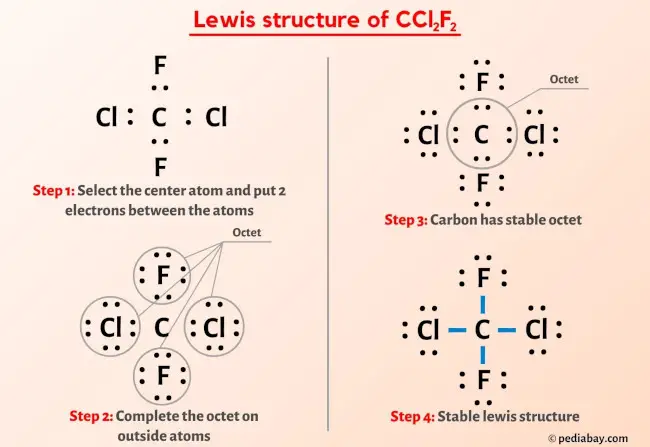
3. Connect the Other Atoms: Attach the two Chlorine (Cl) and two Fluorine (F) atoms to the Carbon with single lines. Each line represents a pair of shared electrons (a covalent bond). So, we've used up 4 pairs of electrons (8 electrons total).
4. Fill the Octets: Now, give each of the surrounding atoms (Cl and F) enough electrons to make them happy. Remember, they want 8 electrons around them (an octet). They already have 2 from the bond with Carbon. So, add 6 more electrons (as three lone pairs of dots) to each Chlorine and each Fluorine.
5. Check Your Work: Double-check that you've used all 28 electrons. If you did it right, Carbon will have 8 electrons around it (4 bonds x 2 electrons per bond), and each Chlorine and Fluorine will also have 8 electrons around them (1 bond x 2 electrons + 3 lone pairs x 2 electrons per lone pair).
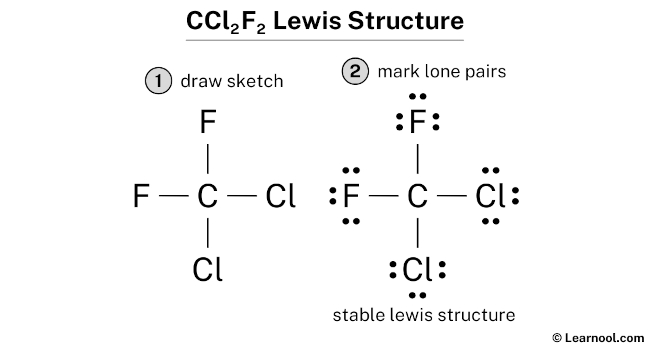
Why Bother?
Okay, so we drew some dots and lines. Why should you care? Because understanding the structure of a molecule tells us a lot about its properties.
The Lewis structure helps us predict how CCl2F2 interacts with other molecules, which is important for understanding its chemical behavior. Remember its impact on the ozone layer? That all started with how CCl2F2 breaks down under UV light, a process we can better understand with its structure.
Think of it like this: knowing how a building is constructed – where the beams and supports are – tells you how strong it is and how it will react to wind and earthquakes. Similarly, the Lewis structure is the blueprint of a molecule, telling us about its strength, stability, and reactivity.
Beyond Freon: The Big Picture
Even though CCl2F2 isn't used as much anymore, the principle of understanding Lewis structures is still incredibly important. It helps us design new materials, develop new drugs, and understand the world around us at a molecular level.
Understanding chemical structures is the foundation of chemistry. It’s how we figure out what things are made of and how they work! From the food we eat to the medicines we take, everything is composed of molecules with specific structures.
So, next time you hear about a chemical formula, don't be intimidated. Remember the Lego analogy, think about building blocks, and you'll be one step closer to understanding the fascinating world of molecules! It's not just about dots and lines; it's about understanding the very fabric of the universe, one molecule at a time. And who knows, maybe you'll even help design the next generation of eco-friendly refrigerants!
