Lewis Structure For Sicl4

Ever doodled atoms and lines? Think of it as molecular art! Let's explore the artistic side of chemistry, specifically looking at SiCl4. It's way more fun than it sounds. Trust me!
Unlocking the Secrets of SiCl4: A Visual Journey
Imagine atoms as LEGO bricks. They want to connect and build something stable. The Lewis structure helps us visualize how these "bricks" link up to form SiCl4, or silicon tetrachloride.
Silicon sits in the middle. Four chlorine atoms surround it. It’s like silicon is the cool kid, and chlorine are the popular kids sitting all around him.
Must Read
Finding the Perfect Fit: Counting Valence Electrons
Now, the fun part. Every atom has a certain number of "connection points." These are called valence electrons. They determine how atoms bond. Think of it as each atom having a certain number of hands to hold.
Silicon has four valence electrons. Each chlorine has seven. We need to add them all up to see the total number of electrons.
So, 4 (from silicon) + 4 * 7 (from chlorines) = 32 valence electrons. That is important. We’ve got 32 "connection points" to work with!
Drawing the Bonds: Sharing is Caring!
We put silicon in the center. Then we surround it with four chlorines. Now draw a line between silicon and each chlorine.

Each line represents a chemical bond, where two atoms are sharing two electrons. Two electrons get a bond.
These bonds represent the connections between silicon and each of the four chlorine atoms. This means we’ve used 8 electrons (4 bonds x 2 electrons/bond).
Completing the Octet: Giving Chlorine its Due
Each atom wants to have eight electrons around it. Except hydrogen which wants two. This is the octet rule. Think of it as an atom's ultimate goal in life!
Silicon already has eight electrons because of the four bonds. Each chlorine only has two. What to do?
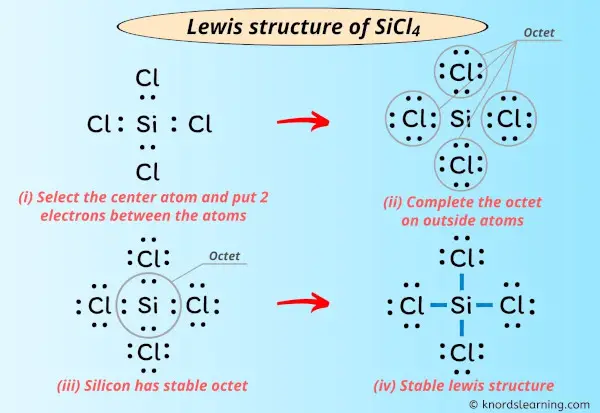
We add lone pairs of electrons to each chlorine atom. These are pairs of electrons that aren't involved in bonding. They're just chilling around the chlorine like comfy blankets.
The Final Masterpiece: SiCl4 Revealed
Each chlorine needs six more electrons (or three more pairs) to complete its octet. So, add three pairs of dots around each chlorine. Voila!
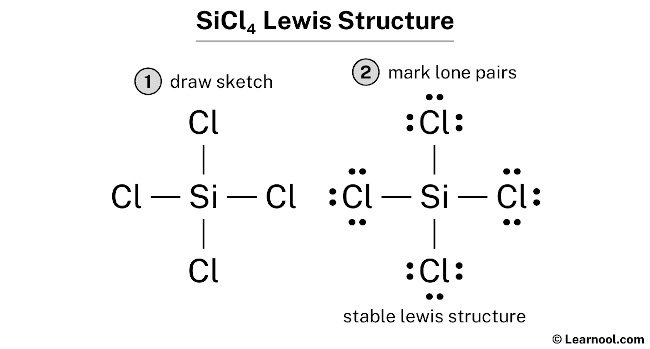
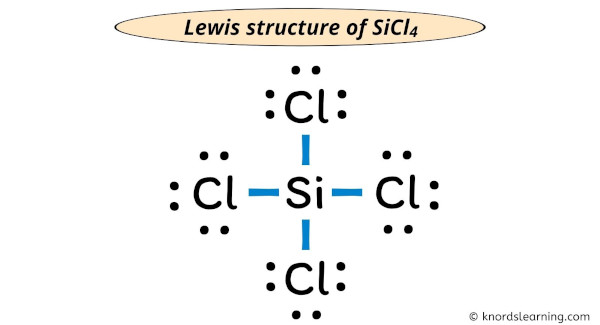
Each chlorine now has eight electrons. Silicon also has eight electrons. Everyone's happy and stable! That is SiCl4 Lewis Structure.
Congratulations. You've just created a Lewis structure for SiCl4. A molecular masterpiece!
Why This Matters: More Than Just Dots and Lines
Lewis structures aren't just pretty pictures. They tell us a lot about a molecule's properties. They help predict how it will react with other molecules. It's like understanding the language of molecules!
The shape of SiCl4 is tetrahedral. This shape influences how it interacts with other substances. It’s a perfect balanced shape, and it is a good example.
These interactions determine everything from a material's strength to its chemical reactivity. It’s important to understand these molecules’ behavior to discover new materials.
Beyond SiCl4: Exploring the Molecular World
SiCl4 is just the beginning. The principles you learned here can be applied to countless other molecules. It opens up a whole new world of chemical possibilities!
The periodic table is full of possibilities. Each element has its own unique bonding behavior. There are so many structures to discover.
Experiment with drawing Lewis structures for different molecules. You might be surprised at what you find. You may even discover new molecules.
So, What’s So Special About SiCl4?
SiCl4 might seem like just another chemical formula. It has a special place in chemistry. It's a perfect example of how atoms bond together to form stable molecules.
Its tetrahedral shape is important in many chemical applications. The molecule is relatively simple. It is perfect for understanding those rules.
Understanding SiCl4 helps us learn more about the world around us. Start your adventure to discover molecular structures. You may fall in love with them!
"The beauty of chemistry lies in the unseen world of atoms and molecules."
Dive into the world of molecular structures. Unleash your inner artist. Explore the endless possibilities that chemistry offers. You might just find your next passion!
