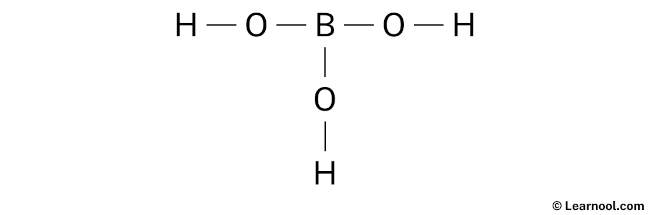
Lewis Structure For H3bo3
Okay, chemistry isn't always the sexiest topic at the dinner table. But stick with me! Today, we're diving into the molecular world of Boric Acid, or H3BO3 as the cool kids (and chemists) call it. Specifically, we're unlocking the secrets of its Lewis structure. Think of it as the VIP backstage pass to understanding how this common compound works. Ready?
Breaking Down Boric Acid: What's the Hype?
Boric acid? You've probably encountered it. It's that mild antiseptic lurking in your first-aid kit, a common ingredient in eye drops, and maybe even a weapon in your battle against unwanted creepy crawlies. But what makes it tick? The answer, my friend, lies in its structure. Knowing the Lewis structure of H3BO3 gives us insight into its properties and behavior. Think of it like knowing the recipe for your favorite dish – suddenly, you understand why it tastes so good.
The Lewis Structure Lowdown: Dots and Dashes
Let's get visual. The Lewis structure is essentially a molecular blueprint. It shows us how the atoms in a molecule are connected and where the electrons are hanging out. It's all about valence electrons, the outermost electrons involved in bonding.
Must Read
Here's the H3BO3 breakdown:
- Boron (B): Boron is the central atom. It has 3 valence electrons. Think of it as the social butterfly of the molecule, always ready to bond, but not with a full eight electrons like most other molecules.
- Oxygen (O): Each oxygen atom has 6 valence electrons. They're looking to share to achieve a stable octet (8 electrons).
- Hydrogen (H): Each hydrogen atom has 1 valence electron. They're easygoing, only needing 2 electrons to be stable.
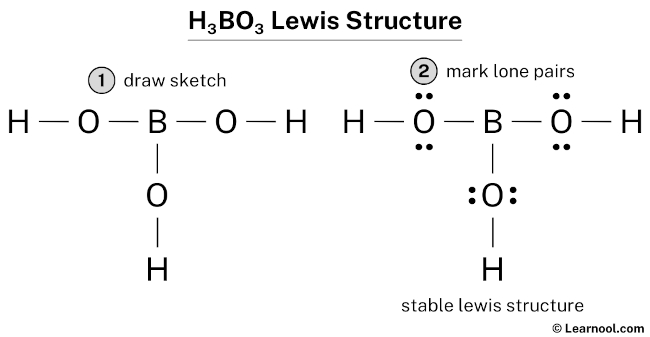
Now, picture this: The boron atom is at the center. Each of the three oxygen atoms is attached to the boron. Each oxygen atom also has a hydrogen atom attached to it. We draw single lines representing shared pairs of electrons (covalent bonds) between the atoms. Boron has three single bonds, one to each oxygen. Each oxygen has one bond to Boron and one bond to hydrogen. Each oxygen atom has two lone pairs of electrons represented by dots to show all of its valence electrons are accounted for.
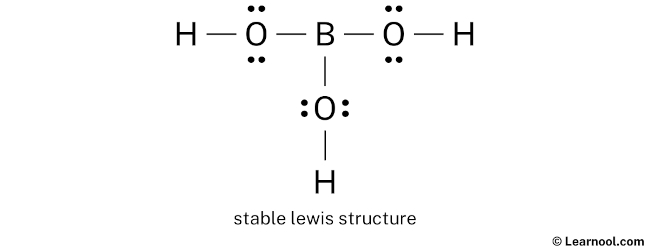
Fun Fact: Boron often doesn't achieve a full octet (8 electrons) in its compounds. It's a bit of a rule-breaker in the chemistry world! This is why boric acid can act as a Lewis acid, accepting electron pairs from other substances.
Drawing Your Own: A Step-by-Step Guide
Feeling ambitious? Let's draw the Lewis structure ourselves:

- Count valence electrons: 3 (B) + 3 * 6 (O) + 3 * 1 (H) = 24 valence electrons.
- Arrange the atoms: Place Boron in the center, with three oxygens surrounding it. Connect each oxygen to the boron. Now add the Hydrogens to each Oxygen.
- Add single bonds: Each bond represents two electrons. Connect each hydrogen to an oxygen and connect each oxygen to the Boron.
- Distribute remaining electrons as lone pairs: We've used 6 single bonds * 2 electrons each = 12 electrons. We have 24 - 12 = 12 electrons remaining. Each oxygen needs 3 lone pairs (6 electrons) to fill its octet. Distribute the remaining 12 electrons as 6 lone pairs on the three oxygen atoms.
Pro Tip: Double-check that each atom (except hydrogen, which only needs two) has a full octet of electrons around it. Boron is a special exception as mentioned earlier!
Beyond the Diagram: Why This Matters
So, you've mastered the Lewis structure. Congratulations! But what does it mean? This structure reveals a few key things:

- Molecular Shape: The arrangement of atoms gives us clues about the molecule's 3D shape.
- Polarity: The uneven sharing of electrons can create a polar molecule, influencing its interactions with other substances.
- Reactivity: Knowing where the electrons are helps us predict how the molecule will react with other chemicals.
Relating to Reality: H3BO3 in Daily Life
Boric acid's properties, revealed by its structure, explain its diverse uses. Its mild acidity and antiseptic properties make it suitable for eye washes and skin treatments. Its ability to disrupt the nervous systems of insects makes it an effective insecticide. Think about it: from soothing irritated eyes to battling household pests, the Lewis structure of H3BO3 plays a crucial role in everyday applications.
Remember: Always use boric acid products as directed. While generally safe in diluted forms, it can be harmful if ingested in large quantities.
The Takeaway: Structure and Significance
Understanding the Lewis structure of H3BO3 isn't just about acing your chemistry exam (though that's a definite perk!). It's about appreciating the elegant connection between molecular structure and real-world applications. Everything, from the medicine we take to the materials we use, is built upon the foundation of atoms and electrons interacting in predictable ways. So, the next time you reach for that bottle of eye drops, take a moment to appreciate the beautifully arranged electrons in the H3BO3 molecule working its magic.