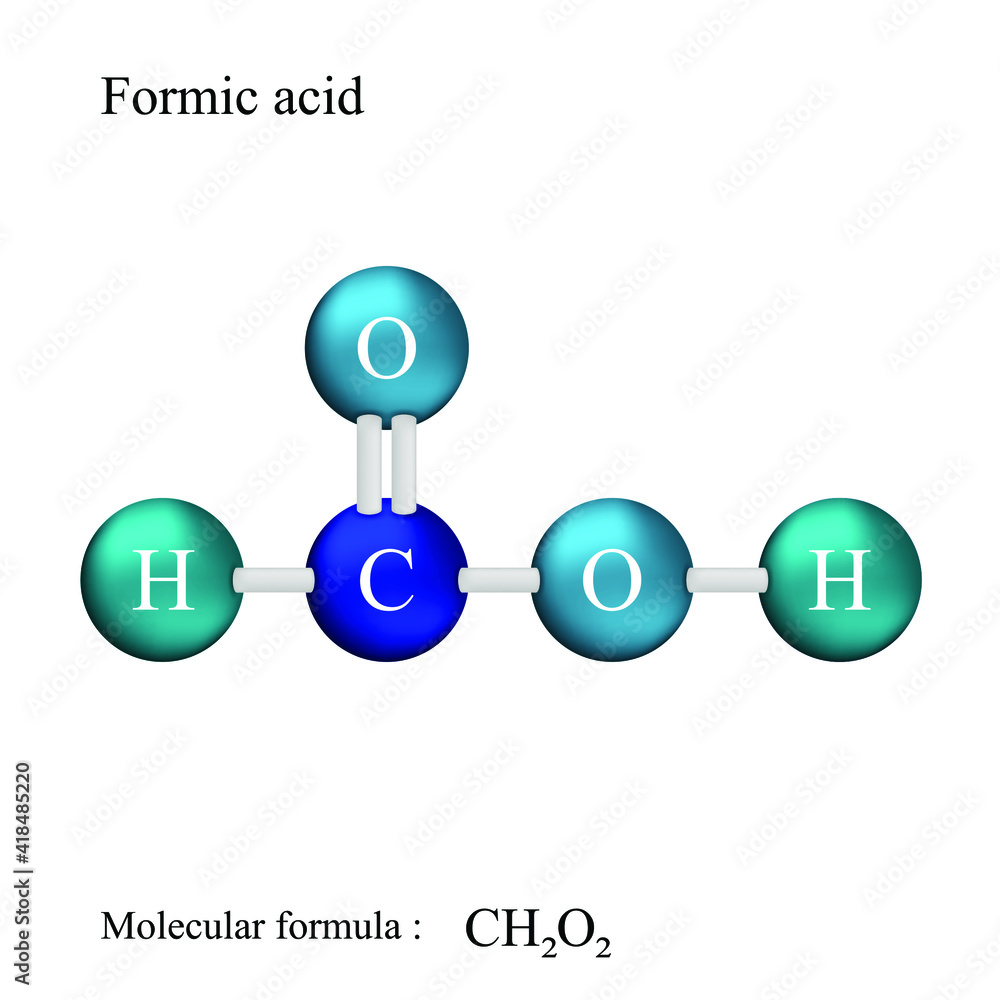
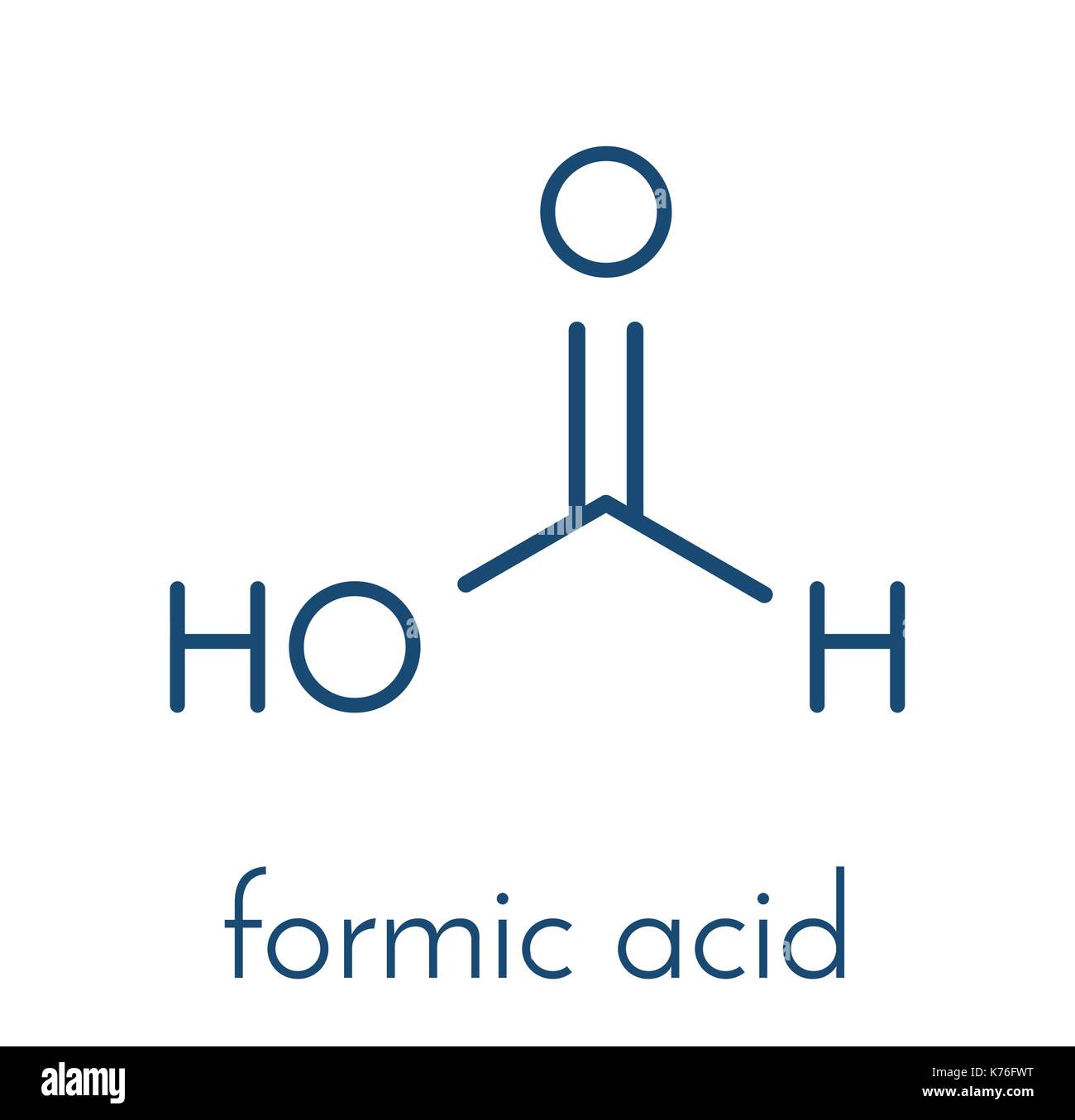
Lewis Structure For Formic Acid
Let's face it, when you hear "Lewis structure," your immediate reaction probably isn't a burst of excitement. But stick with me! Think of Lewis structures as the ultimate visual aid for understanding how molecules are built. They're like the instruction manuals for the tiny, invisible world around us. Why do people actually enjoy drawing them? Because they unlock secrets! They allow you to predict how substances will behave, which makes understanding chemistry, and the world, much easier.
So, why bother drawing the Lewis structure for something like formic acid? Well, formic acid (also known as methanoic acid) is that stuff that makes ant bites sting. Understanding its structure helps us understand why it stings! More broadly, Lewis structures are incredibly beneficial because they allow us to predict a molecule's shape, its polarity (whether it's slightly positive or negative), and its reactivity (how likely it is to react with other substances). This has huge implications for everyday life.
Think about it: drug design. Pharmaceutical companies use Lewis structures to design molecules that will bind to specific targets in the body, allowing them to create new and improved medicines. Or consider material science. Understanding the structure of polymers (long chains of molecules) allows scientists to create stronger, more flexible plastics. Even in cooking, understanding how molecules interact (partly based on their Lewis structures) helps you predict how ingredients will combine and react, leading to tastier dishes! Understanding the structure of formic acid specifically helps us understand why it's a good antibacterial agent, often used in livestock feed.
Must Read
Now, let's get practical and talk about drawing the Lewis structure for formic acid (HCOOH). First, count the valence electrons: Hydrogen has 1, Carbon has 4, and Oxygen has 6. So, 1 + 4 + (2 * 6) + 1 = 12 valence electrons total. Carbon is usually the central atom (though in this case, it’s a bit more complex due to the two oxygens). Start by drawing single bonds between the atoms: H-C-O-H, with another oxygen branching off the carbon. Now, distribute the remaining electrons as lone pairs around the oxygen atoms to satisfy the octet rule (8 electrons around each atom except for hydrogen, which only needs 2). You'll find you're short! This means you'll need to form a double bond between the carbon and one of the oxygen atoms. You'll end up with a structure where the carbon has a single bond to one oxygen (which also has a hydrogen attached) and a double bond to the other oxygen.
To enjoy Lewis structures more effectively, remember these tips: Practice makes perfect! Start with simple molecules and work your way up to more complex ones. Always double-check your electron count and ensure each atom (except hydrogen) has a full octet. Use different colors to represent electrons from different atoms – it can help prevent confusion. And finally, don't be afraid to use online resources and tutorials! There are plenty of excellent visual aids available to guide you. The more comfortable you become with Lewis structures, the more you'll appreciate their power in explaining the chemical world around you!