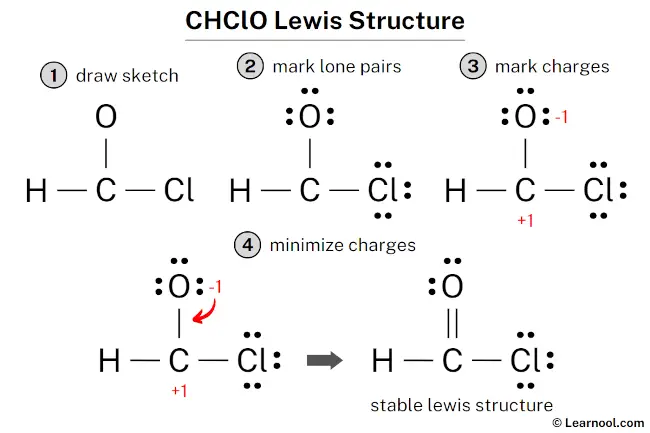
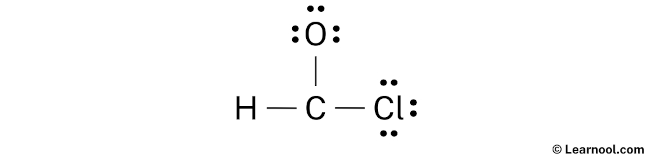
Lewis Structure For Chclo
Let's talk chlorine monoxide, shall we? Or, as the cool kids call it: ClO. It’s a simple molecule, right? Chlorine, oxygen… what could go wrong?
Drawing the Line (Structure, That Is!)
Time for a Lewis structure! Remember those? You’re probably having flashbacks to high school chemistry right now. Don’t worry, I am too.
First, we count valence electrons. Chlorine brings 7. Oxygen brings 6. That’s 13 total. Thirteen electrons to arrange around our Cl and our O. Hmmm. Already feels like a Friday the 13th situation.
Must Read
So, you slap those atoms together. Cl-O. Boom. Bonded. Easy peasy. Now, we fill in the octets. Oxygen gets six more electrons (three lone pairs). Chlorine gets six more electrons (three lone pairs as well). Everyone's happy, right? Wait. Something's nagging me.
That nagging thing? That’s the odd number of electrons. 13. You can't exactly evenly distribute 13 electrons among two atoms and call it a day. One atom has to have less than an octet.

Here comes the "unpopular opinion" part. Are you ready for it?
I think we overcomplicate ClO.
Seriously! We spend so much time agonizing about formal charges and who gets the radical (the unpaired electron). We draw resonance structures until our pencils break.

The Radical Dilemma
The radical. Dun dun DUN! One atom gets stuck with it. We debate whether it's better for chlorine or oxygen to be the "odd one out."
Some folks passionately argue that oxygen, being more electronegative, should be happier with the negative formal charge. Therefore, Chlorine should have the radical. Others say chlorine, being bigger and more polarizable, can handle the stress of the unpaired electron better. It's like choosing between the lesser of two evils… which, frankly, is most of life anyway.
And the formal charges! Oh, the formal charges. Is it better to minimize them, even if it means breaking the octet rule on one atom? Is it better to obey the octet rule and live with a slightly larger formal charge? It's a chemical philosophical quandary!
My "Radical" Solution (Pun Intended)
Here's my take. It's a radical! It’s unstable! It wants to react. Why are we trying to make it conform to our neat and tidy rules? It's like trying to put a wild animal in a tiny cage.
Maybe, just maybe, the best Lewis structure for ClO is the one that acknowledges its chaotic nature. A slightly messy structure, perhaps. A structure that screams, "I'm not perfect! I'm unstable! I'm going to react with something!"
Okay, okay, I know. That's not very scientific. It doesn't give you neat bond orders or predictable formal charges. But isn't there some beauty in accepting the inherent messiness of the universe? Especially when it comes to molecules that desperately want to break the rules?

So next time you're struggling to draw the "perfect" Lewis structure for ClO, take a deep breath. Remember it's a radical. Let it be a little messy. Embrace the chaos.
And maybe, just maybe, draw a little smiley face next to the electron dot on the less fortunate atom. Just to let it know you empathize with its plight. Hey, we've all been there.
Ultimately, it's all just dots and lines representing how scientists think this molecule probably looks most of the time. It's an educated guess. But don't let perfect be the enemy of good enough when you’re just trying to get by.
