Lewis Structure For Ch2cl2
Okay, let's talk about something thrilling: drawing the Lewis structure for CH2Cl2. Buckle up, buttercups!
The Great CH2Cl2 Adventure
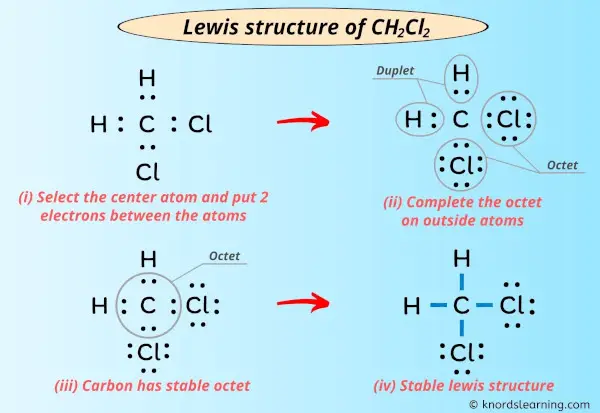
First, we've got carbon (C). Carbon's like that friendly neighbor who always wants to share. It needs four bonds to be happy.
Then we have hydrogen (H). Hydrogen is super simple. It only needs one bond. One and done, baby!
Must Read
And finally, chlorine (Cl). Chlorine is a bit needier, wanting just one bond but loaded with lone pairs. Think of it as that friend who always carries extra snacks "just in case."
Putting the Puzzle Together
So, we have one carbon, two hydrogens, and two chlorines. How do we make them all play nice?
Carbon needs four bonds. Let's stick those hydrogens and chlorines onto it like decorative (slightly reactive) ornaments.
Essentially, the carbon is at the center, like the star of our show. Hydrogen and chlorine surround it. It's a molecular mosh pit of bonding!
The "Unpopular" Opinion (Brace Yourselves)
Here's my hot take. You ready? I think drawing every. single. lone. pair. is... overrated.

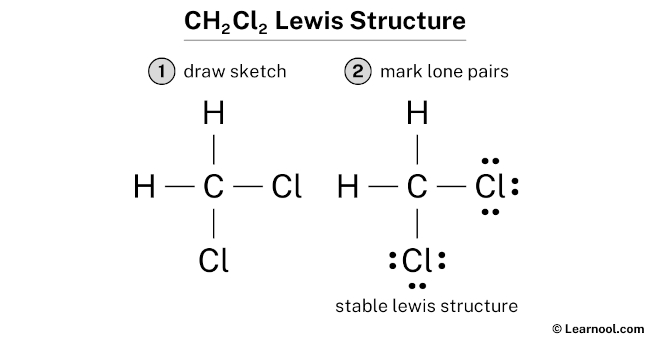
I know, I know! The horror! But hear me out. We know chlorine has those extra electrons. We know it's got its little electron cloud happening.
Do we really need to draw six little dots around each chlorine every single time? It just feels...cluttered.
The Case for (Slightly) Lazy Lewis Structures
Think about it. We're not dummies. We understand the principles. We get the octet rule.
Sometimes, a cleaner, more streamlined structure just helps visualize things better. It's like decluttering your closet – suddenly, you can see what you have!
Okay, okay, I'm not saying abandon all best practices. I'm just suggesting a little... selective laziness.
Visualizing the Mayhem
Imagine the carbon in the middle. A hydrogen atom is bonded to it. Another hydrogen also attached to the carbon.

Then, a chlorine swings in and bonds, and another chlorine on the other side. Bonds everywhere!
It's not a line; it's a party! A tetrahedron party, to be exact, because CH2Cl2 is tetrahedral in shape. Remember that shape, it's important.
The Grand Finale (and a Confession)
So, there you have it. My (slightly heretical) take on drawing the Lewis structure for CH2Cl2.
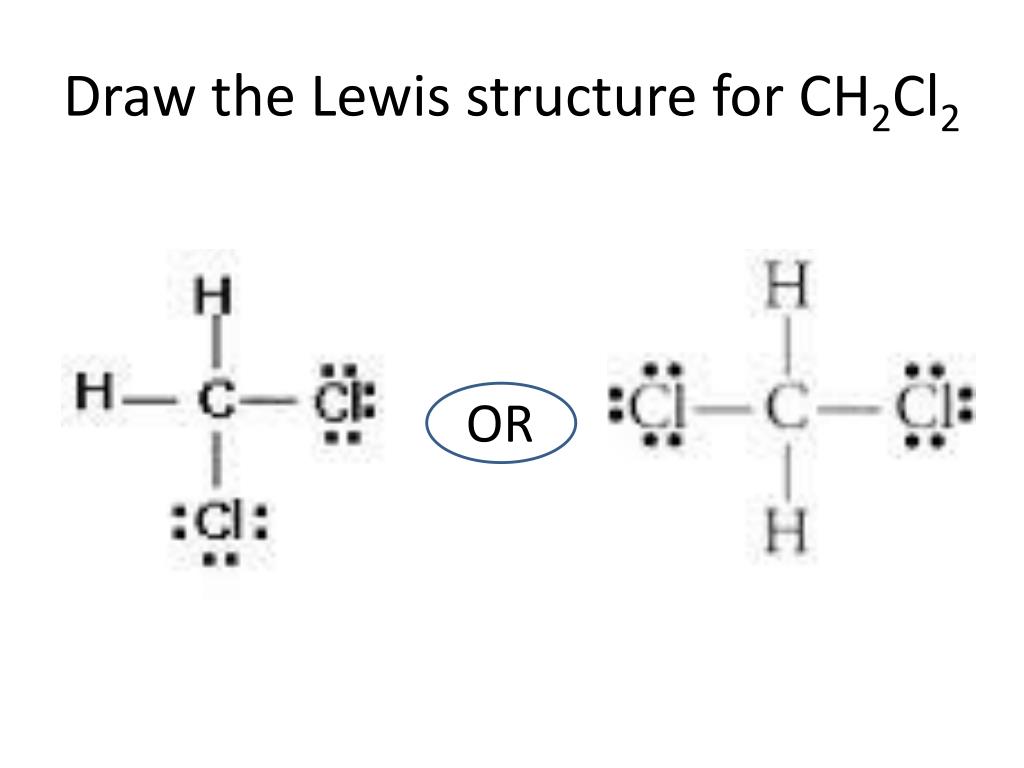
Draw those lone pairs if you must! It's your Lewis structure; you do you.
But maybe, just maybe, consider the benefits of a slightly more... minimalistic approach. Let's call it "artistic license."
And now, a confession. Sometimes, when I'm feeling particularly rebellious, I don't even draw the bonds as lines. I just write "C-H-H-Cl-Cl" and hope for the best. Don't tell my chemistry professor.
In Conclusion (Sort Of)
CH2Cl2. It's a molecule. It has a Lewis structure. And it's probably used in something important somewhere. Who knew?
The important thing is that you now know (or at least have been exposed to) the general idea.
Now go forth and draw some molecules! Or, you know, don't. The choice is yours.
Just remember my unpopular opinion when you get tired of drawing dots. Let that be your mantra! Be free!
Remember, chemistry is about understanding the concepts, not just becoming a human dot-drawing machine.
So, embrace the beauty (and the occasional frustration) of chemistry. And maybe, just maybe, give those lone pairs a little break now and then.
Good luck and have fun with CH2Cl2! May your Lewis structures be ever in your favor.
And always remember this: double-check everything, because even though this is all said in jest, real chemistry has real consequences.
Don't blow up your lab, okay?
Finally, if you forget what a Lewis structure for CH2Cl2 looks like, just draw something that vaguely resembles it and hope no one notices. You're welcome!
