Lewis Structure For Cf2cl2

Okay, let’s talk CFCs. Specifically, that notorious troublemaker: CF2Cl2. It's not the catchiest name, I know.
My Unpopular Opinion on Drawing Molecules
Let’s be honest. Drawing these things can feel like a pop quiz you didn't study for. It’s all lines and dots.
Some people make it look SO easy. Are they wizards? Probably.
Must Read
The Dance of the Electrons
Think of it like this: every atom is trying to be popular. They all want a full "octet" – eight electrons in their outer shell. It’s like trying to get the most likes on social media. Everyone’s striving for validation.
Carbon, in the middle of CF2Cl2, is your master networker. It needs four connections to be happy.
Each fluorine and chlorine only need one.
Let's Draw!
First, put carbon (C) smack-dab in the center. It's the star of our show.

Then, surround it with two fluorine (F) and two chlorine (Cl) atoms. Try to space them out evenly. We want a symmetrical vibe, you know?
Now, let's add those dots! Fluorine and chlorine bring seven electrons each to the party. They only need ONE to feel complete.
Lone Pairs: The Wallflowers
These are electrons that aren't bonding. They're just chilling, not making connections.
Fluorine will have three pairs of these lone electrons. Chlorine too. Don’t forget them!
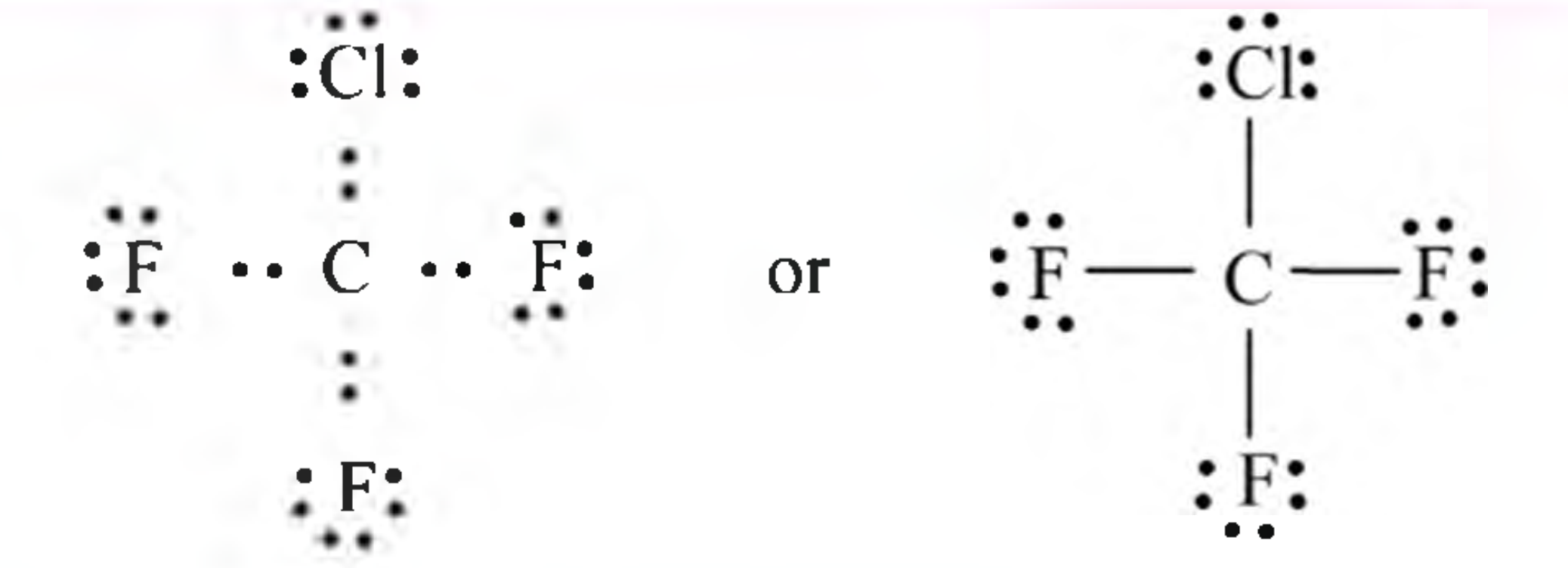
Think of lone pairs as the shy kids at the dance. They're there, but they're not exactly cutting a rug. But they’re important! Everyone needs a bit of personal space.
The Lines That Bind (and Sometimes Break the Ozone)
Each line represents a shared pair of electrons. This is where the atoms hold hands. A chemical bond is formed.
Carbon is holding hands with each fluorine and each chlorine. It's fully booked! No more electron dates for you, carbon.
Each fluorine and each chlorine is also happy. They have eight electrons buzzing around them.
The Final Product (Simplified!)
Okay, so you’ve got C in the middle. Two lines going to F and two lines going to Cl. Each F and Cl has six dots around it.
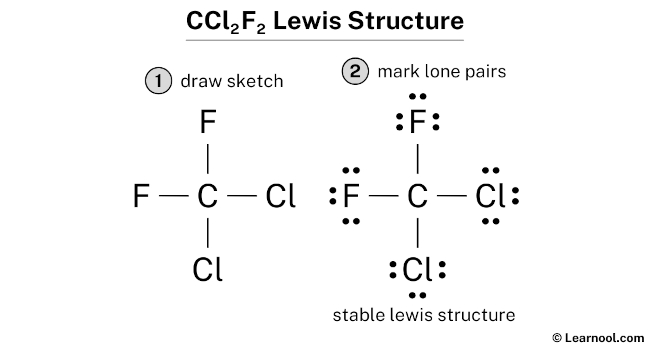
Voila! You've (basically) drawn the Lewis structure for CF2Cl2.
It's not perfect. I'm not perfect. But we got the main idea down, right?
Why This Matters (A Little Bit)
So, why even bother with all this dot-and-line madness? Because this little molecule caused big problems.
CF2Cl2 used to be a common refrigerant. But it turns out, when it floats up to the stratosphere, it breaks down. And releases... chlorine.

And chlorine is NOT a friend to the ozone layer. So CF2Cl2 was banned! It’s like that one party guest who always causes drama. You just have to politely ask them to leave.
My Controversial Conclusion
Drawing Lewis structures might seem tedious. But understanding how molecules bond is pretty important. Especially when those molecules are messing with our planet!
So, next time you see a chemical formula, don’t run screaming. Take a deep breath. And remember the dance of the electrons!
And maybe, just maybe, you'll start to see the beauty (and sometimes the danger) hidden in those little lines and dots.
Learning chemistry doesn't have to be dull. It's all about finding the humor in the madness!
