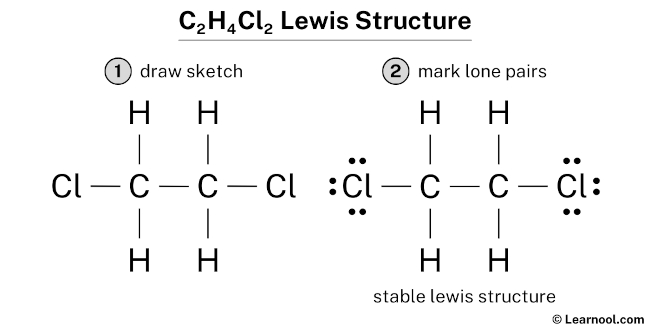
Lewis Structure For C2h4cl2

Ever wonder how scientists visualize molecules, those tiny building blocks that make up, well, everything? It's not like they're looking at them with super-powered microscopes and sketching what they see. Instead, they use tools like Lewis structures, which are a bit like molecular blueprints. And today, we're going to dive into one specific molecule: C2H4Cl2, also known as dichloroethane. Why? Because understanding Lewis structures is surprisingly useful, and it’s a fun way to peek into the world of chemistry.
So, what exactly is a Lewis structure? It’s a diagram that shows the connectivity of atoms within a molecule, along with all the valence electrons. These are the outermost electrons that participate in chemical bonding. Think of it as a visual guide to how atoms share electrons to form a stable molecule. The purpose of a Lewis structure is to help us understand a molecule’s bonding, its shape, and even some of its properties. For example, knowing the structure can give clues about how a molecule will react with other substances.
Why is this useful? Well, in education, Lewis structures are foundational. They’re taught in introductory chemistry courses to build a strong understanding of bonding concepts. Students use them to predict molecular geometries, understand polarity, and even predict reaction mechanisms. But it's not just for academics! Understanding these structures can help you decipher ingredient lists in cleaning products or even understand how medications interact with your body on a molecular level. Imagine seeing a chemical name and being able to visualize, even roughly, how it's put together. That's empowering!
Must Read
Consider C2H4Cl2. There are two main isomers: 1,1-dichloroethane and 1,2-dichloroethane. Drawing the Lewis structures for each reveals their different connectivity. For 1,1-dichloroethane, both chlorine atoms are attached to the same carbon. For 1,2-dichloroethane, each chlorine atom is attached to a different carbon. These seemingly small differences can significantly impact the molecule's properties. For example, different isomers can have different boiling points or reactivities.
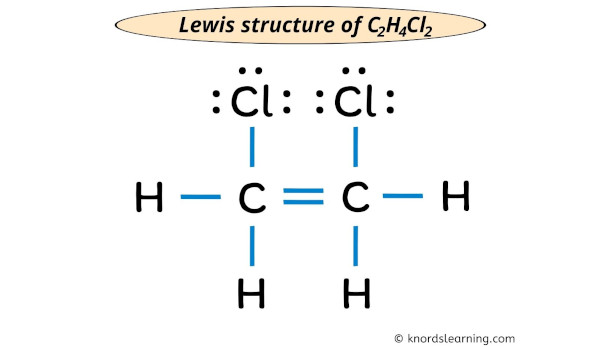
Now, how can you explore this further? Grab a piece of paper and a pencil (or your favorite digital drawing tool!). Start by figuring out the total number of valence electrons in C2H4Cl2. Carbon has 4, hydrogen has 1, and chlorine has 7. So, (2 x 4) + (4 x 1) + (2 x 7) = 26 valence electrons. Then, sketch the atoms, connecting them with single bonds. Remember, carbon usually forms four bonds. Distribute the remaining electrons as lone pairs around the chlorine atoms and any remaining atoms until each atom (except hydrogen, which is happy with two) has an octet (eight) of electrons around it. If you run out of electrons before everyone has an octet, consider forming double or triple bonds. Don't be afraid to experiment and look up examples online for guidance. There are plenty of resources available, including interactive simulations that let you build and manipulate molecules. Sites like Chem LibreTexts or Khan Academy offer excellent tutorials and practice problems.
The key is to practice. Start with simple molecules like water (H2O) or ammonia (NH3) and gradually work your way up to more complex ones like C2H4Cl2. You'll be surprised at how quickly you start to "see" molecules in your mind's eye. So, grab your pencil, embrace the challenge, and unlock the secrets of the molecular world. Happy drawing!