Lewis Dot Structure For Pf3
Alright, chemistry curious minds, let's dive into something that sounds intimidating but is actually quite elegant: the Lewis Dot Structure for PF3. Think of it as the molecular equivalent of sketching a quick floor plan before you rearrange your furniture. It’s all about visualizing how atoms connect and share their precious electrons.
Why Bother with Dots?
Before we get dotted, let's understand why we even bother. Lewis Dot Structures, named after the brilliant Gilbert N. Lewis (a name you should casually drop at your next trivia night), are simplified diagrams that show the valence electrons – the outermost electrons – of atoms and how they bond together. These structures help us predict a molecule's shape, polarity, and reactivity. Basically, it's molecular matchmaking!
PF3: Our Star Molecule
Our star for today is PF3, or Phosphorus Trifluoride. Sounds fancy, right? It's a colorless gas used in some chemical reactions (don't try this at home, folks!). Now, let's break down how to draw its Lewis structure, step-by-step.
Must Read
Step 1: Counting the Valence Electrons
First things first: the electron census. We need to count the total number of valence electrons in PF3. Phosphorus (P) is in Group 15 (or 5A) of the periodic table, meaning it has 5 valence electrons. Fluorine (F) is in Group 17 (or 7A), giving it 7 valence electrons. Since we have three fluorine atoms, we multiply 7 by 3, getting 21. Adding the 5 from phosphorus, we have a grand total of 26 valence electrons to work with.
Step 2: The Central Atom
Next up, deciding who takes center stage. Usually, the least electronegative atom goes in the middle. Fluorine is a notorious electron hog, so Phosphorus takes the central position. Think of it like hosting a dinner party: the least demanding guest gets to choose the main course.

Step 3: Single Bonds, Please
Now, let's connect each fluorine atom to the phosphorus atom with a single bond. Each bond represents a shared pair of electrons. So, we’ve used up 2 electrons per bond, meaning we've used 3 bonds * 2 electrons/bond = 6 electrons. That leaves us with 26 - 6 = 20 electrons to distribute.
Step 4: Completing the Octets
Fluorine, being a polite guest at this electron party, wants to achieve a full octet – 8 valence electrons. We start by placing lone pairs of electrons around each fluorine atom until each has 8 electrons in total. Each fluorine already has 2 from the bond, so we need to add 6 more, or 3 lone pairs. That's 3 fluorines * 6 electrons/fluorine = 18 electrons used. We have 20 - 18 = 2 electrons left.
Step 5: The Lone Pair on Phosphorus
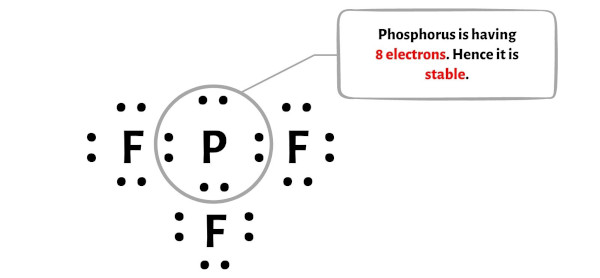
Those last two electrons? They go on the phosphorus atom as a lone pair. This completes the Lewis structure! Phosphorus now has one lone pair and three single bonds, meaning it effectively has 8 electrons around it (6 from the bonds + 2 from the lone pair). Octet rule achieved (for phosphorus, at least)!
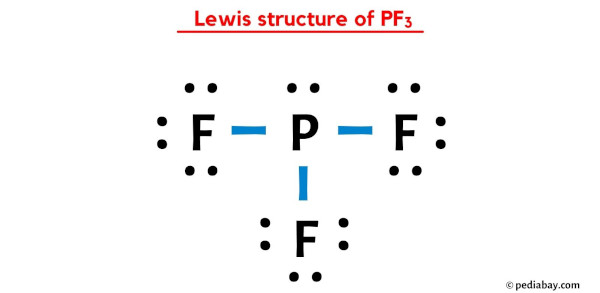
The Final Picture
So, the Lewis Dot Structure for PF3 looks like this (imagine it described because we can't draw it here directly, but visualize it!): P in the center, bonded to three F atoms, each F having three lone pairs of electrons, and P having one lone pair.
Molecular Geometry: A Little Bonus
Because of that lone pair on the phosphorus, PF3 isn't perfectly symmetrical. It has a trigonal pyramidal shape. Think of it like a pyramid with a triangular base. This shape is crucial for understanding how PF3 interacts with other molecules. Shape matters, even in the molecular world. It's like knowing the right handshake.

Fun Fact Interlude
Did you know that the concept of electron pairing, which is at the heart of Lewis structures, has influenced fields beyond chemistry? Its philosophical implications, relating to duality and balance, have even seeped into some artistic interpretations and psychological theories. Who knew electron dots could be so profound?
Practical Tips for Lewis Structure Success
- Always double-check your electron count. A miscount can throw the whole structure off.
- Be patient. Sometimes it takes a few tries to get it right. Think of it as a puzzle.
- Practice, practice, practice! The more you do, the easier it becomes. Start with simple molecules and work your way up.
From Dots to Daily Life
You might be thinking, "Okay, cool dots, but how does this relate to my life?" Well, understanding Lewis structures, and more broadly, how atoms interact, helps us understand the world around us. From the medicines we take to the materials our clothes are made of, everything is built on the principles of chemical bonding. It allows us to design new materials, understand biological processes, and even mitigate environmental problems. Knowledge of fundamental concepts such as this can influence how we perceive the world and make decisions within it.
Furthermore, the process of solving a Lewis structure problem, breaking it down into smaller steps, is a valuable skill that translates to problem-solving in everyday life. Just like PF3, complex problems often become manageable when you break them down and tackle each element individually. So, embrace the dots! They're more than just dots; they're a window into the intricate and fascinating world of chemistry.

