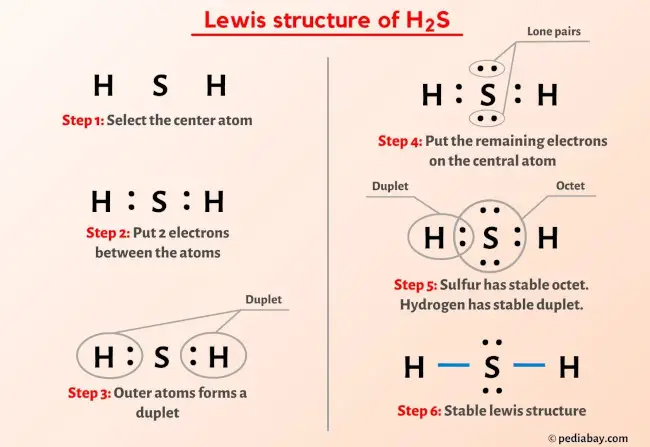
Lewis Dot Structure For H2s
Alright, let's dive into the wacky world of molecules! Today's star? Hydrogen sulfide, or as I like to call it, H2S – the molecule with a nose for trouble! Get ready because we're about to draw its Lewis Dot Structure.
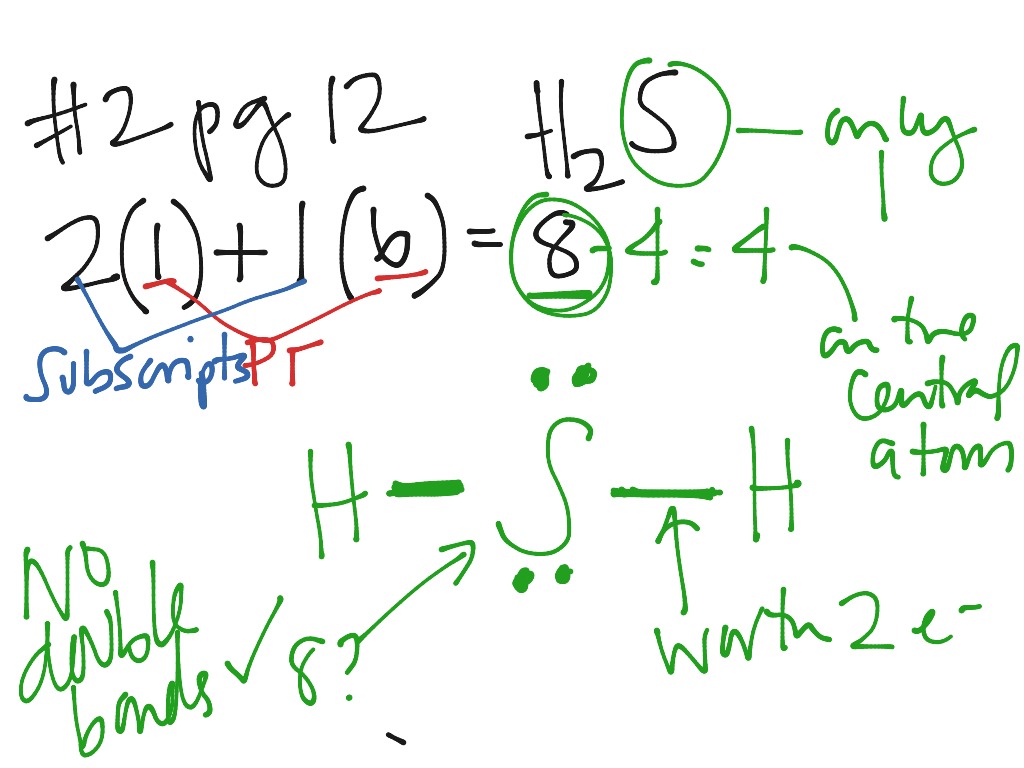
First things first, we need to wrangle the electrons. Think of electrons like tiny, energetic puppies, eager to pair up and cause a little... bonding! Each atom brings its own puppy to the party.
Hydrogen's Lone Wolf Electron
Hydrogen (H) is a pretty simple guy. He's got one electron, a single, solitary puppy, ready to mingle.
Must Read
Since we have two hydrogens in H2S, that's a total of 2 electrons from Team Hydrogen. Consider them the enthusiastic but slightly clueless newcomers.
Sulfur's Six-Pack of Electrons
Now, sulfur (S) is a bit more complex. Sulfur struts in with a whopping six electrons, like a seasoned pro at the electron dance.
Sulfur is in the same family on the periodic table as oxygen (think water, H2O), so its electron situation is similar, just a little bigger and smellier (metaphorically!). Remember oxygen, it's sulfur's cool cousin.
Electron Tally: The Grand Total
Time to add up all the electron puppies! Two from the hydrogens, and six from sulfur.
That gives us a grand total of eight electrons to play with when drawing our Lewis Dot Structure. Eight little dots of bonding brilliance!
The Central Atom Showdown: Sulfur Takes the Stage
So, who gets to be the star of the show, the central atom around which everything else orbits? It's usually the least electronegative element (besides hydrogen), or the one that needs to form the most bonds. That's sulfur.
Picture sulfur as the cool kid in school that everyone wants to be friends with. Hydrogen? Well, they're just happy to be included!
Bonding Bonanza: Hydrogen Clings to Sulfur
Now for the fun part: drawing the bonds! Remember, a bond is just a shared pair of electrons, like two puppies becoming best friends and deciding to share a bone.
We draw a line from each hydrogen to the sulfur atom. Each line represents two shared electrons, a covalent bond, a match made in electron heaven!
Dots, Dots Everywhere: Completing the Octet
Sulfur started with six electrons. Each bond with hydrogen gives it two more. But sulfur is only surrounded by 4 electrons (2 bonds). Since sulfur wants 8, we have to add in lone pairs.
A lone pair is a pair of electrons that aren't involved in bonding, but still belong to the atom. Think of them as sulfur's personal stash of electrons, like a secret candy hoard.
So we draw two pairs of dots on the top and bottom of the sulfur. These represent the 4 unbonded electrons.
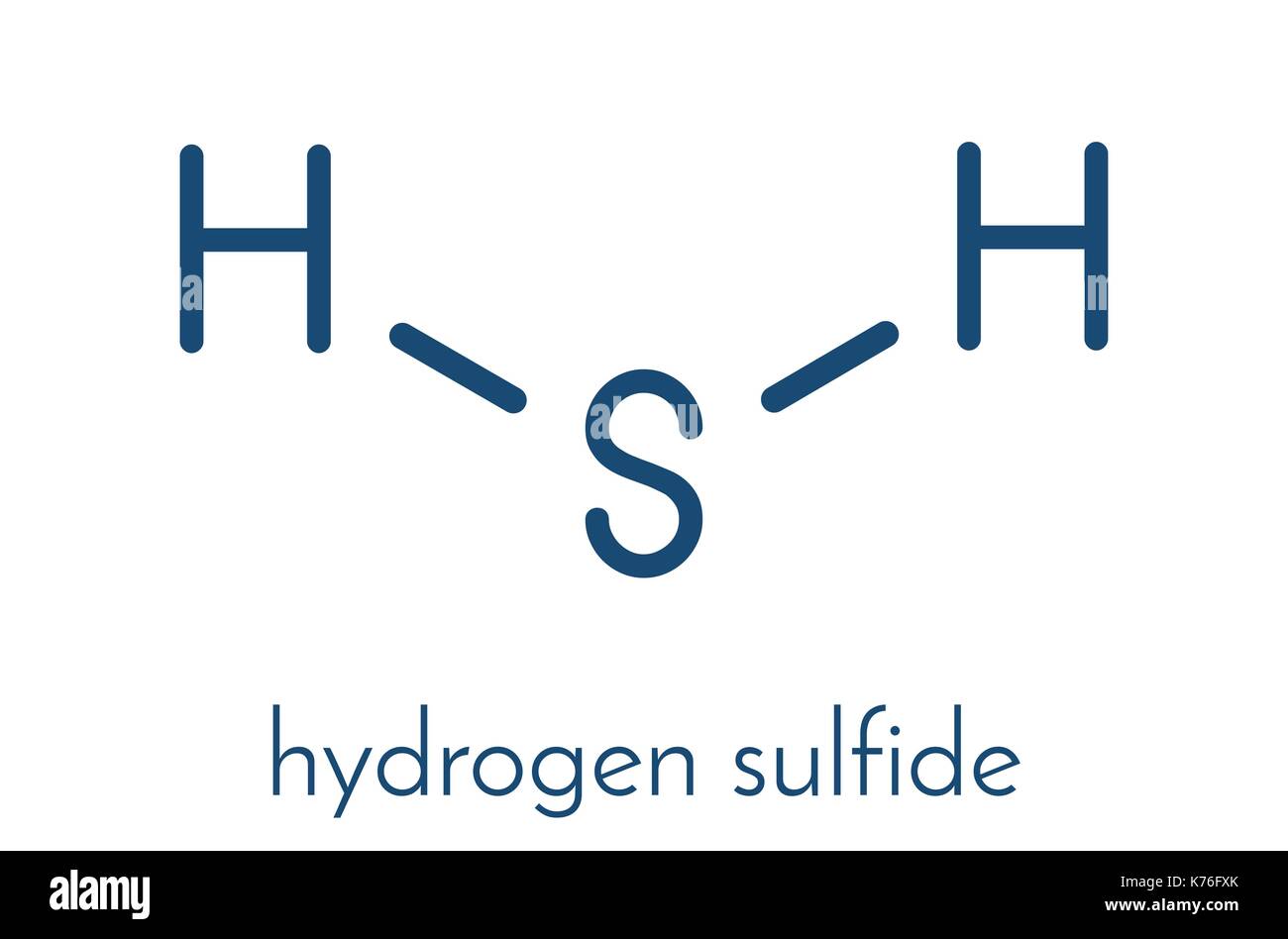
Ta-Da! The Lewis Dot Structure of H2S
And there you have it! The Lewis Dot Structure of H2S is complete! Sulfur in the middle, bonded to two hydrogens, and sporting two lovely lone pairs of electrons. A magnificent molecular masterpiece, if I do say so myself!

It looks something like this (imagine dots for the lone pairs and lines for the bonds): H-S-H .. ..
Why This Matters: Unveiling Molecular Secrets
Why bother with all these dots and lines? Because the Lewis Dot Structure helps us understand how molecules behave! It gives us clues about their shape, polarity, and reactivity.
For instance, the two lone pairs on sulfur influence the shape of the molecule, making it bent rather than linear. This bent shape contributes to H2S's polarity. The molecule isn't symmetrical, which influences how it interacts with other molecules.
Polarity Party: H2S's Slightly Negative Side
Because sulfur is more electronegative than hydrogen, it pulls the shared electrons in the bonds closer to itself. This creates a slightly negative charge on the sulfur atom and a slightly positive charge on each hydrogen atom.
It's like a tug-of-war where sulfur is winning, creating a charge imbalance in the molecule. This polarity affects how H2S interacts with other molecules, especially water.
Smelling Trouble: H2S and Our Noses
Now, let's address the elephant in the room (or rather, the rotten egg smell). H2S is notorious for its pungent odor, a characteristic that's hard to forget. This smell also affects how it bonds with some metals.
Our noses are incredibly sensitive to H2S, able to detect even tiny concentrations. It's a useful adaptation, as high concentrations of H2S can be harmful.

Beyond the Basics: H2S in the Real World
H2S isn't just about bad smells and chemistry lessons. It's a molecule with real-world implications, both good and bad.
It's found in volcanic gases, sewage, and natural gas. It's also used in various industrial processes, from producing sulfuric acid to synthesizing certain chemicals.
H2S: A Cautionary Tale (and a Chemical Wonder)
While H2S can be useful, it's important to remember its toxicity. It can be harmful to breathe in high concentrations and can even be fatal.
So, treat it with respect, handle it with care, and always work in a well-ventilated area. Chemistry is fascinating, but safety is paramount!
Lewis Dot Structures: The Big Picture
Learning about Lewis Dot Structures isn't just about memorizing rules and drawing dots. It's about understanding the fundamental principles of chemical bonding and molecular structure.
It's about visualizing the invisible world of atoms and electrons and appreciating the intricate dance that creates the molecules around us. By following this example, we can apply the same knowledge to more complex compounds.
So, Keep Exploring!
So, keep exploring the world of chemistry! There are countless molecules out there waiting to be discovered, each with its own unique properties and stories to tell. The more Lewis Dot Structures you draw, the better you'll become at understanding the language of molecules!
Remember, chemistry is all about experimentation, exploration, and a little bit of fun. So, grab your pencil, your periodic table, and your imagination, and get ready to unleash your inner chemist!
From Dots to Discoveries: The Journey Continues
And that, my friends, is the story of the Lewis Dot Structure of H2S! From humble hydrogen atoms to stinky sulfur, we've uncovered the secrets of this fascinating molecule, one dot at a time.
Remember, even the most complex molecules can be understood by breaking them down into their simplest components and following the rules of electron sharing. Now go forth and conquer the chemical world, one dot at a time!
A Final Word of Encouragement
Don't be afraid to make mistakes, to ask questions, and to challenge yourself. The beauty of chemistry lies in its ability to surprise and delight, to reveal the hidden wonders of the universe. Keep your mind open and be ready to learn! And don't forget to appreciate the amazing world around you.
Every molecule, from the simplest to the most complex, has a story to tell. It's up to us to listen, to observe, and to unravel the mysteries that lie within. So, embrace the challenge, celebrate your successes, and never stop learning! And with that, go practice your Lewis Dot Structure skills!
Embrace Your Inner Electron Whisperer
And that, my friends, concludes our electron adventure for today! Keep practicing, keep exploring, and never lose your sense of wonder. With practice, understanding the Lewis Dot Structure will become second nature.
Now go forth and create some molecular masterpieces. Chemistry is waiting!
