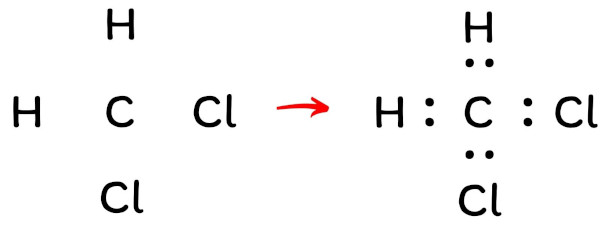
Lewis Dot Structure For Ch2cl2
Hey there, chemistry adventurer! Ever stared at a chemical formula like CH2Cl2 and thought, "Right, what do I do with that?" Don't worry, you're not alone! Let's break down how to draw the Lewis Dot Structure for dichloromethane (that's CH2Cl2 in fancy chemical speak). It's easier than you think, I promise! (Easier than parallel parking, anyway.)
Step 1: Count Those Valence Electrons!
First things first, we need to know how many valence electrons we're dealing with. Remember, valence electrons are the ones on the outermost shell of an atom – they're the rockstars that do all the bonding.
- Carbon (C) has 4 valence electrons.
- Hydrogen (H) has 1 valence electron each. Since we have two, that's 2 x 1 = 2 valence electrons from the hydrogens.
- Chlorine (Cl) has 7 valence electrons each. And guess what? We have two chlorines! So, 2 x 7 = 14 valence electrons from the chlorines.
Add 'em all up: 4 + 2 + 14 = 20 valence electrons in total. That's our magic number. We have 20 little dots to place strategically. Think of it like a valence electron scavenger hunt!
Must Read
Step 2: Pick a Central Atom (It's All About Location, Location, Location!)
Now, who gets to be the star of the show? In CH2Cl2, it's carbon. Carbon likes to be in the center because it can form the most bonds (it's a social butterfly like that). Hydrogen and chlorine are more like wallflowers and tend to hang out on the edges.
Step 3: Connect the Dots (Literally!)
Alright, let’s hook everything up. Carbon is in the middle, and we'll connect it to the two hydrogens and the two chlorines with single bonds.

Each single bond represents two electrons. So, we've used 4 bonds x 2 electrons/bond = 8 electrons so far. That leaves us with 20 - 8 = 12 electrons to play with. The suspense is killing me!
Step 4: Fulfilling the Octet Rule (Everyone Wants Eight!)
The octet rule is a fancy way of saying that most atoms want to be surrounded by eight electrons to be stable. Hydrogen is the exception; it only wants two (it's happy with a duet). Carbon already has its eight electrons from those four bonds. Lucky carbon!
Now, let's focus on the chlorines. Each chlorine is currently sharing one pair of electrons with the carbon atom, meaning each chlorine only has two electrons. To satisfy the octet rule, we need to add three more pairs of electrons (six more electrons) to each chlorine atom. BOOM!
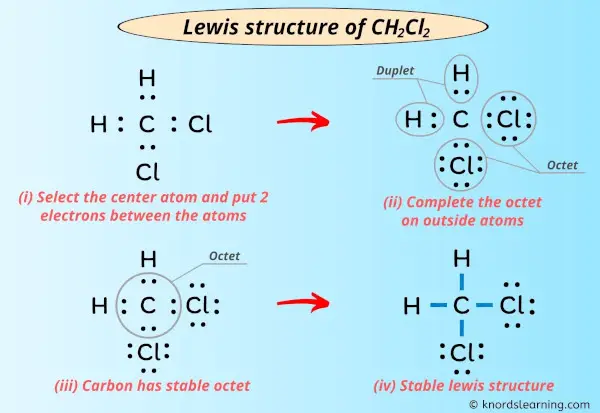
So, we add six electrons around each chlorine, arranged as three lone pairs. 6 electrons x 2 chlorine = 12 electrons. And guess what? We used all 12 electrons we had remaining! Perfect. No leftovers!
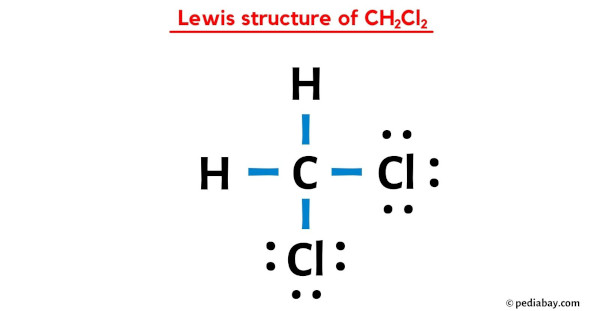
Step 5: Behold! The Lewis Dot Structure
At this point, you should have a carbon atom in the center bonded to two hydrogen atoms and two chlorine atoms. Each chlorine atom should have three pairs of dots around it (those are the lone pairs). And that, my friend, is the Lewis Dot Structure for CH2Cl2!

If you’re feeling extra fancy, you can use lines to represent the bonds instead of drawing out all the dots, that will give you a structural diagram.
Quick Recap and Helpful Tips
- Count all valence electrons.
- Pick a central atom. Usually, it's the least electronegative or the one that can form the most bonds.
- Connect atoms with single bonds.
- Complete the octet (or duet for hydrogen) by adding lone pairs.
- Double-check that you've used the correct number of valence electrons.
Remember, drawing Lewis structures is like piecing together a puzzle. It takes practice, but it gets easier with time. Don't be discouraged if you don't get it right away! Just keep practicing, and soon you'll be drawing Lewis Dot Structures in your sleep! (Okay, maybe not in your sleep, but you'll be pretty good at it!)
So, go forth and conquer those chemical structures! With a little bit of patience and a whole lot of enthusiasm, you'll be a Lewis Dot Structure master in no time. And who knows, maybe you'll even impress your friends at your next chemistry-themed party! You got this!