Lewis Dot Structure For C2h4
Ready for a little molecular matchmaking? We're diving into the wonderful world of Lewis Dot Structures, and this time, our star is C2H4, also known as ethene! Think of these structures as little blueprints for molecules, showing us how atoms get together and share their stuff.
Forget boring textbooks! We're going to look at how C2H4, that unassuming little molecule, uses its dots to create something pretty cool. Imagine each dot is a tiny electron, eager to pair up and form a bond. It's like a singles mixer, but for atoms!
The Players: Carbon and Hydrogen
First, let's introduce our contestants: Carbon (C) and Hydrogen (H). Carbon's a bit of a social butterfly. It needs four partners (electrons) to feel complete. Hydrogen, on the other hand, is happy with just one. It's the easygoing friend everyone loves.
Must Read
Now, how do we figure out how these two play together? That's where the fun begins! We're going to draw some dots. Carbon starts with four lonely dots around it, representing its four valence electrons. Each hydrogen brings along its own single dot. Think of them as tiny handshakes waiting to happen.
Drawing the Dot Diagram: It's a Molecular Dance!
Here's the tricky (but awesome) part. We need to arrange these atoms so that everyone's happy. Carbon wants four bonds, and Hydrogen wants one. How do we achieve this harmonious arrangement?
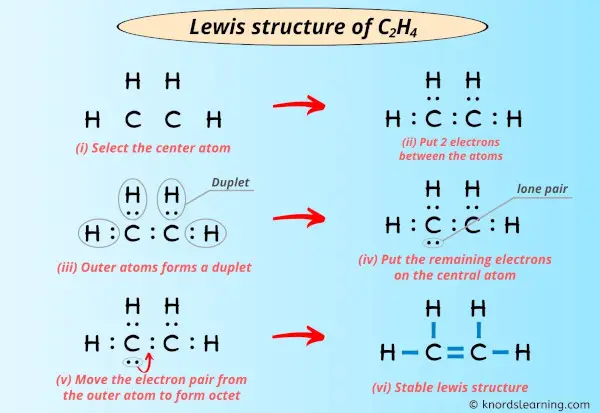
Well, two carbon atoms decide to get cozy. They're going to share not just one, but two pairs of electrons. That's right, a double bond! It's like holding hands really, really tight. Each carbon then grabs two hydrogen atoms, forming single bonds with them. Everyone gets what they need, and a stable molecule is born. It’s a beautiful thing, really!
So, when you draw your Lewis Dot Structure for C2H4, you'll see two carbons connected by a double bond. Each carbon will also be connected to two hydrogens by single bonds. And all those dots? They'll be neatly paired up, showing which atoms are sharing their electrons.

Why is this so Entertaining?
You might be thinking, "Dots? Bonds? What's so exciting about that?" Trust me, it's more engaging than it sounds! Lewis Dot Structures let you visualize the invisible. You're seeing, in a simplified way, how molecules are built. It's like being an architect, designing structures one bond at a time.
Plus, they give you clues about how molecules will behave. The number of bonds, the types of bonds – all these things influence a molecule's properties. So, understanding these structures is like having a secret decoder ring for the chemical world.
More Than Just Dots: It's Understanding Chemistry
Think about it: C2H4 is the stuff that ripens bananas and helps make plastics. By understanding its Lewis Dot Structure, you're getting a glimpse into the chemistry behind everyday things. It's not just abstract science; it's relevant to the world around you.
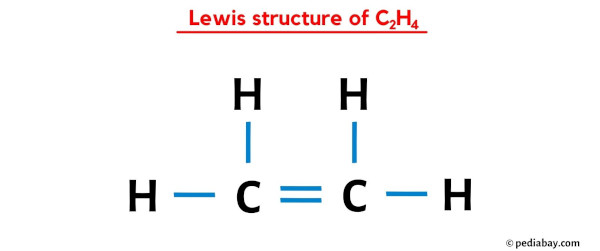
It's also surprisingly satisfying when you get it right! When all the dots are paired up and everyone's happy, you've successfully built a molecule on paper. It’s a small victory, perhaps, but a victory nonetheless.
Don't be afraid to get your hands dirty (figuratively, of course) and try drawing some Lewis Dot Structures yourself. Start with simple molecules like water (H2O) or methane (CH4). The more you practice, the better you'll get at understanding how atoms connect and share their electrons.
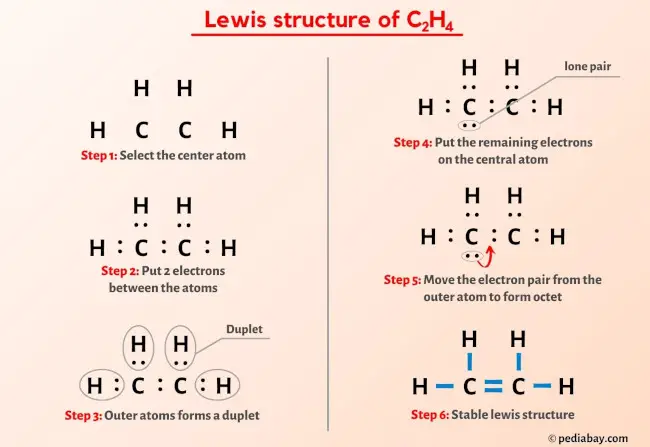
And who knows? Maybe you'll even start seeing the world in a whole new way – as a collection of molecules, all interacting and bonding according to the laws of chemistry. It's a perspective that's both fascinating and incredibly rewarding.
So, grab a pencil and some paper and get ready to explore the wonderful world of Lewis Dot Structures. C2H4 is just the beginning! There's a whole universe of molecules out there waiting to be discovered, one dot at a time.
Remember: Chemistry isn't just about memorizing facts; it's about understanding how things work at a fundamental level. And Lewis Dot Structures are a fantastic tool for doing just that.
Now, go forth and bond!
