Lewis Dot Structure For Bf3
Okay, let's talk about Boron Trifluoride, or as the cool kids call it, BF3.
Specifically, let's talk about its Lewis Dot Structure.
Brace yourselves, because I have an unpopular opinion.
Must Read
The BF3 Situation
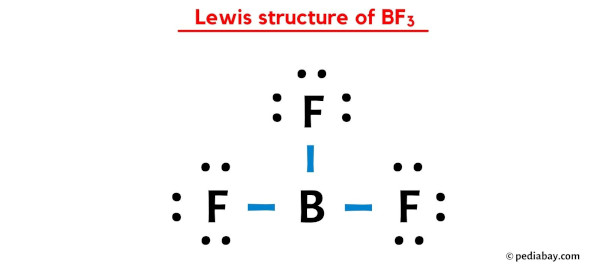
We all know the "official" Lewis Dot Structure. Boron in the center. Three fluorines surrounding it. Each fluorine happily sporting its octet.
Boron? Well, Boron gets six electrons. It's "electron deficient," they say.
And everyone’s just…okay with that? Really?
My Problem With It
Look, I get the rules. I really do.
But something about that lone Boron, sitting there with only six electrons, just rubs me the wrong way.
It looks…incomplete. Like a puzzle missing a piece. Or a pizza missing two slices.

I know, I know, Boron is an exception. It's happy with less.
But is it really happy? Or is it just pretending to be so it doesn't have to admit it made a mistake?
I bet if we could ask Boron, it would secretly want that full octet.
Let's Get Controversial
Now, here’s where I get into trouble.
What if we, just for fun, pretended Boron could form a double bond with one of the fluorines?
Hear me out!
Yes, I know Fluorine is electronegative. Yes, I know it "doesn't like" to form double bonds.

But what if, just this once, it made an exception for its buddy Boron?
Imagine the possibilities!
Suddenly, Boron has its octet. Everyone is happy! Fluorine gets to show off its bonding prowess. Peace reigns in the molecular kingdom.
Okay, maybe not everyone is happy.
Your chemistry professor probably isn’t thrilled with my suggestion.
Why This Makes Sense (To Me, At Least)
Think about resonance structures. We draw them all the time to represent molecules where electrons are delocalized.
So, why can't we imagine BF3 as a resonance hybrid, where one of the structures includes that sneaky double bond?
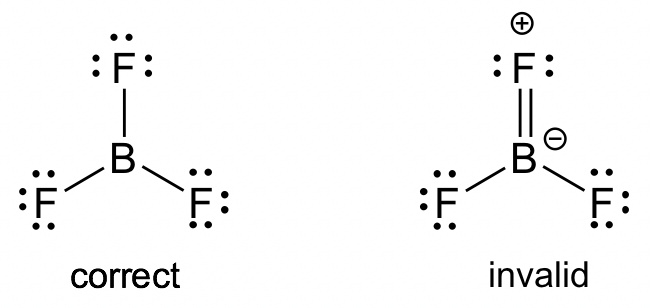
Maybe it’s not a major contributor, but it's there, lurking in the background, giving Boron a little extra electron love.
Plus, it explains BF3's behavior a little better, doesn’t it?
It's a strong Lewis acid, meaning it loves to accept electron pairs. Why?
Because it’s secretly yearning for that full octet, even if it pretends it doesn't.
The Point Is...
I'm not saying the accepted Lewis Dot Structure for BF3 is "wrong."
I'm just saying… maybe it's not the whole story.
Maybe, just maybe, there’s a little bit of double-bonded Fluorine lurking in the shadows, giving Boron a helping hand.

So, next time you draw the Lewis Dot Structure for BF3, spare a thought for that lonely Boron.
And maybe, just maybe, draw a little dotted line suggesting a possible double bond.
Your professor might not appreciate it, but Boron certainly will.
In conclusion, sometimes breaking the rules (a little bit) can help us understand things better.
Or, at the very least, it makes chemistry a little more interesting.
Thank you for coming to my TED Talk.
"The problem with the world is that intelligent people are full of doubts, while stupid people are full of confidence." - Bertrand Russell
I am clearly in the first camp when it comes to BF3. Good luck on your next chemistry exam!
