Is Sugar An Element Mixture Or Compound

Ever baked a cake and wondered what exactly makes it so… sweet? Or maybe you've seen "sugar-free" labels and pondered what's being taken out? Understanding what sugar is is surprisingly fascinating and relevant to our everyday lives. It's not just about satisfying a sweet tooth; it's about understanding basic chemistry and how the world around us is built. Let's dive in and see if sugar is an element, a mixture, or a compound. Trust me, it's sweeter than you think!
So, what’s the big deal? Why should we care about the composition of sugar? Well, for starters, sugar is a fundamental source of energy for our bodies. We use it to power our brains, muscles, and pretty much everything in between. Understanding its structure can help us make informed choices about our diet and the impact of different types of sugars on our health. From a purely academic standpoint, it’s a brilliant example of how elements can combine to form complex molecules with unique properties. It’s a building block for understanding more complex chemistry.
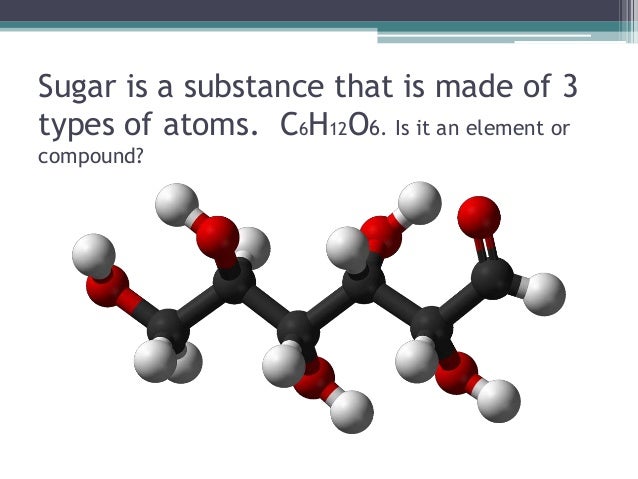
Alright, let's get down to it. Sugar, in its most common form (like the table sugar you sprinkle on your cereal), is called sucrose. Sucrose isn’t a single element. It's also not a mixture. A mixture is a combination of substances that are physically combined but not chemically bonded, like salad dressing or air. You could, in theory, separate the components of a mixture relatively easily.
Must Read
Sugar is a compound. That means it's made up of two or more different elements that are chemically bonded together in a fixed ratio. In the case of sucrose, it consists of three elements: carbon (C), hydrogen (H), and oxygen (O). Its chemical formula is C12H22O11. This precise arrangement of atoms gives sugar its unique sweet taste and other properties. Think of it like building with LEGOs; carbon, hydrogen, and oxygen are the individual bricks, and sucrose is the specific structure you build with them.
Where do we see this in action? Well, in education, understanding sugar's composition is a classic chemistry lesson. Students learn about chemical formulas, covalent bonds, and how different elements interact. In daily life, think about reading nutrition labels. Recognizing that "sugar" isn't just one thing, but a compound, allows you to appreciate the different types of sugars (glucose, fructose, etc.) and their potential effects on your body.

Want to explore this further? Here are a few simple tips: Next time you're baking, pay attention to how sugar dissolves in water. This is a physical change, but it illustrates how the sugar molecules interact with water molecules. You can also try comparing the sweetness of different types of sugars, like honey (which contains fructose and glucose) versus table sugar. Or, look up the molecular structure of other common compounds, like water (H2O) or salt (NaCl), and compare them to sucrose. It's all connected!
Understanding that sugar is a compound, and not an element or mixture, opens a door to understanding so much more about the chemical world around us. So, go ahead, embrace your inner scientist and explore the sweet world of chemistry!