Is Nitrogen A Inner Transition Metal

Hey there! Ever wonder about the cool building blocks that make up, well, everything? Let's dive into the wild world of elements and tackle a quirky question.
Nitrogen: The Life of the (Party?) Element
We all know nitrogen. It's in the air we breathe! About 78% of the atmosphere is nitrogen gas.
It's also super important for plants. Think fertilizer! Plants need it to grow big and strong.
Must Read
So, Is it an Inner Transition Metal? Hold on to Your Hats!
That's the million-dollar question, isn't it? You might be picturing shiny, reactive metals. The type that sometimes make a splash. Think of the elements at the very bottom of the periodic table!
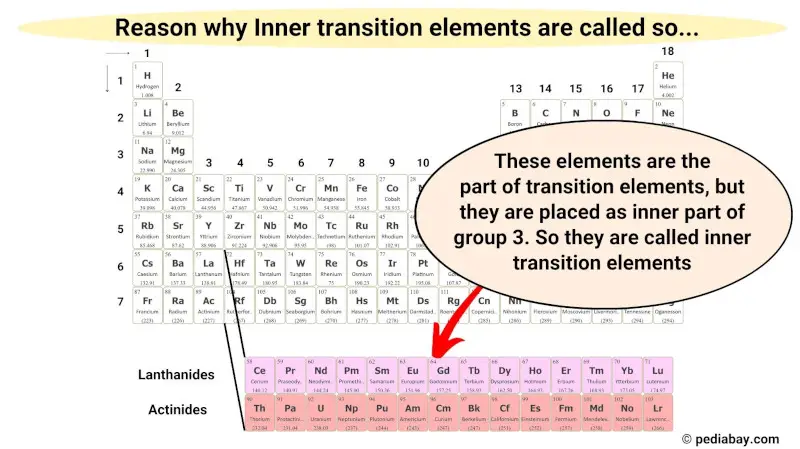
Inner transition metals? They're the rockstars of the metal world. Tucked away at the bottom of the periodic table.
These elements, also called the lanthanides and actinides, are known for their unusual electron configurations and properties.
They're all about filling up their inner electron shells. That's what gives them their special characteristics. Think glow-in-the-dark stuff, nuclear reactors, and super strong magnets.
The Verdict: Nitrogen's Place in the Periodic Table
Nitrogen? An inner transition metal? The short answer is a resounding no!
It's a nonmetal. Completely different ballpark.
Nitrogen hangs out much higher up on the periodic table. Specifically in Group 15, also known as the nitrogen group.
It is on the right side of the table. Nowhere near the inner transition metal block.
Why the Confusion? Probably Because Chemistry is Awesome!
So, why might someone even ask if nitrogen is an inner transition metal? Probably because chemistry has a lot of exceptions.
The periodic table is complex! There are a ton of details that are easy to miss.
And sometimes, elements do things that surprise us! It is not a metal.
Nitrogen's Nonmetal Personality
Nitrogen prefers to form gases. It's a building block for amino acids and DNA.
It's a key part of the molecules that make up life. Super different from the inner transition metals!
Think of it this way: nitrogen is a social butterfly. It bonds with many elements.
It forms stable molecules like N2 (the air we breathe). It also makes less stable, explosive compounds, like nitroglycerin.
Inner transition metals, on the other hand, are often more focused on their inner selves. Busy with those inner electron shells!

More Fun Nitrogen Facts
Did you know liquid nitrogen is incredibly cold? It can freeze things instantly!
It's used in cryogenics. Like preserving biological samples.
It even makes ice cream super smooth!
Inner Transition Metals: The Real Deal
Let's give the inner transition metals some love! Elements like uranium and plutonium are part of this group.
They're known for their radioactive properties. They're used in nuclear power.
And in some very important research. They're found at the bottom of the periodic table.
Elements like lanthanum and cerium are also inner transition metals. These are used in electronics. As well as catalysts.
They are super important in modern life. But in very different ways than nitrogen.

Comparing and Contrasting: Why Nitrogen Isn't Like Them
The key difference lies in electron configuration. It decides how the element will behave.
Inner transition metals have partially filled f orbitals. Nitrogen is all about those p orbitals.
This difference in electron configuration dictates their chemical properties. This also defines their role.
Exploring the Periodic Table: A Never-Ending Adventure
So, nitrogen isn't an inner transition metal. But that's okay!
It has its own unique role to play. It's an essential part of life.
Think of it as the quiet hero. Working behind the scenes.
Dive Deeper: Resources to Learn More
Want to explore more about elements? The periodic table?
Check out educational websites. There are lots of interactive resources out there.

You can find videos and articles. They make learning chemistry fun!
In Conclusion: Embrace the Quirks of Chemistry!
The world of chemistry is full of surprises! It can be confusing. But it can be awesome!
Don't be afraid to ask questions. Explore the periodic table.
You might just discover your favorite element. Keep exploring and stay curious!
So, next time someone asks you if nitrogen is an inner transition metal, you can confidently say, "Nope! But it's still super cool!"
A Final Thought: The Importance of Asking Questions
Asking questions, even if they seem silly, is how we learn. It is also how we grow.
Never stop being curious about the world around you. Especially the elements that make it up!
Who knows what amazing discoveries you'll make? Keep the curiosity alive!
