Is Nacl Hydrophobic Or Hydrophilic
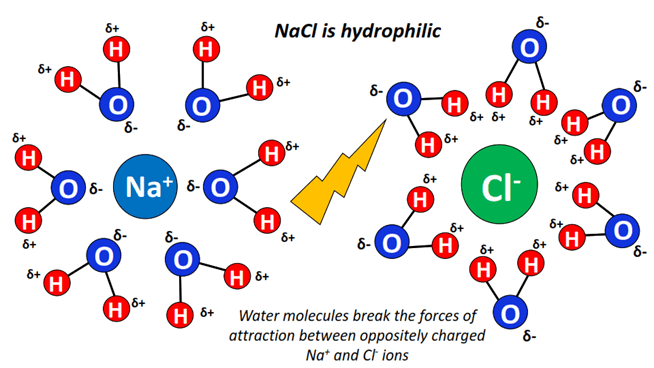
Hey there! Ever wondered if your table salt, you know, good ol' NaCl, is more of a "water-lover" or a "water-hater"? It's a surprisingly interesting question, right?
Let's dive in! So, is NaCl hydrophobic (afraid of water) or hydrophilic (loves water)? Drumroll, please...
It's definitely hydrophilic! No contest. But why, you ask? Good question! Let's break it down.
Must Read
Think of water molecules. They're like tiny magnets, seriously. They have a slightly positive end and a slightly negative end. We call it polarity. Fancy, huh?
Now, NaCl is an ionic compound. That means it's made up of ions – positively charged sodium ions (Na+) and negatively charged chloride ions (Cl-). Opposites attract, remember from science class?
So, when you toss salt into water (like, say, when you're making pasta – yum!), the water molecules literally swarm around the sodium and chloride ions. They use their positive and negative ends to latch onto the ions. It's like a molecular dance party!
This whole process is called dissolving or hydration. The water molecules effectively pull the ions apart, breaking the ionic bonds that hold the salt crystal together. Poof! The salt disappears! (Well, it doesn't really disappear, but you know what I mean.)

But what if salt was hydrophobic? Imagine that! You'd sprinkle salt on your pasta and it would just sit there, stubbornly refusing to dissolve. The water would be all, "Ew, no way, not touching that!" Disaster!
Why is it so important to be hydrophilic anyway?
Glad you asked! Our bodies rely on this whole dissolving-in-water thing big time! We need salt (in moderation, of course!) for all sorts of bodily functions – nerve impulses, muscle contractions, you name it. And how does it get around our system? Dissolved in water (blood, mostly!).
If NaCl were hydrophobic, it wouldn't dissolve properly and our bodies wouldn't be able to absorb it. Talk about a problem! We'd be a seriously dehydrated and salty mess. (Okay, maybe not that dramatic, but still… bad news.)

So, just to recap: water is polar, salt is ionic, and opposites attract. That attraction leads to dissolving, which is essential for life! Pretty cool, huh?
Let's contrast this with something that is hydrophobic, like oil. Ever tried mixing oil and water? They separate, right? That's because oil molecules are non-polar. They don't have those positive and negative ends that water can latch onto. They're like the wallflowers at the molecular party – all, "Nah, we're good on our own."
In short, salt's hydrophilic nature is due to its ionic structure and the polar nature of water. They're a match made in molecular heaven! It's why your tears are salty, why the ocean is salty, and why your pasta water… well, should be salty! (Don't forget the salt, people!)
So next time you sprinkle salt on your food, remember that you're witnessing a tiny miracle of chemistry. You're watching hydrophilic salt happily dissolve in the water, bringing flavor and life to everything it touches! Who knew salt could be so exciting?
Now, if you'll excuse me, I think I need a salty snack. Maybe some pretzels? Or potato chips? For science, of course!
