Is F2 Paramagnetic Or Diamagnetic

Hey there, science buddy! Ever wondered about the hidden lives of molecules? Specifically, F2, also known as fluorine? Let's dive into a seriously cool question: Is F2 paramagnetic or diamagnetic? It's more exciting than you think!
What's the Deal with Magnetism, Anyway?
First, a quick primer. Imagine tiny bar magnets within molecules. Okay, they're not really tiny bar magnets, but electrons act like them! Electrons have spin. Think of it like they're constantly twirling. This spin creates a tiny magnetic field.
If all the spins are neatly paired up – one spinning "up" and the other "down" – their magnetic fields cancel out. Boom! Diamagnetism. These molecules are repelled by a magnetic field. Kind of shy, right?
Must Read
But, if there are unpaired electrons kicking around, causing a magnetic moment? The molecule gets attracted to a magnetic field. Hello, paramagnetism! These guys are magnetic extroverts!
So, unpaired electrons equal paramagnetic. Paired electrons equal diamagnetic. Simple, right?
Fluorine: The Wild Child of the Halogens
Fluorine. It's the most reactive element. It's a total drama queen. But what about its magnetic personality?
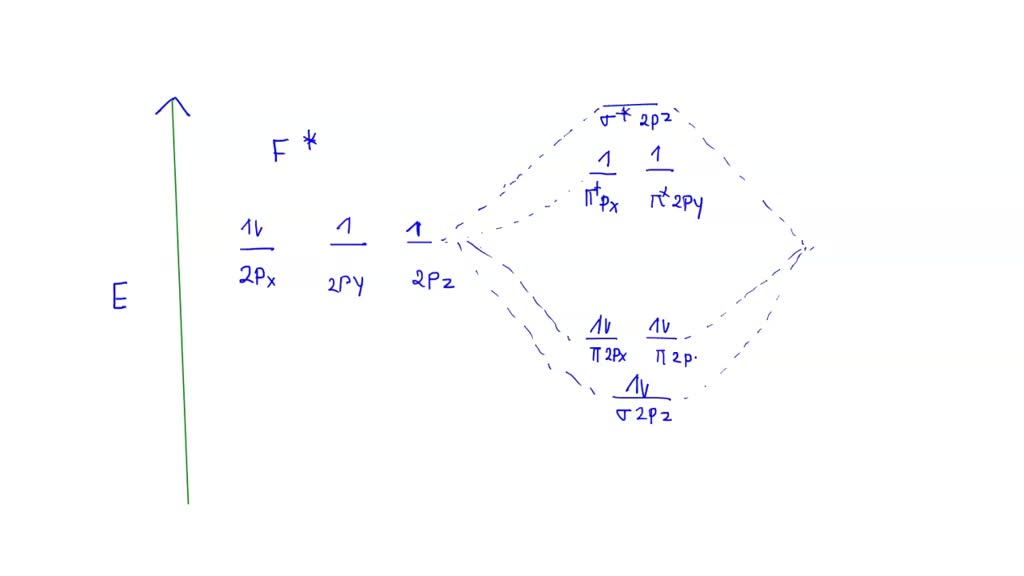
You might think, "Okay, let's just look at the molecular orbital diagram!" And you’d be right… eventually! This diagram shows how atomic orbitals combine to form molecular orbitals in F2. These orbitals have different energy levels, and we fill them up with electrons.
Now, if you look at the simple molecular orbital diagram of O2, you'll notice two unpaired electrons in the pi antibonding orbitals. Those unpaired electrons are the reason O2 is paramagnetic!
Many people assumed that the same would be true for F2!
However... sneaky fluorine!
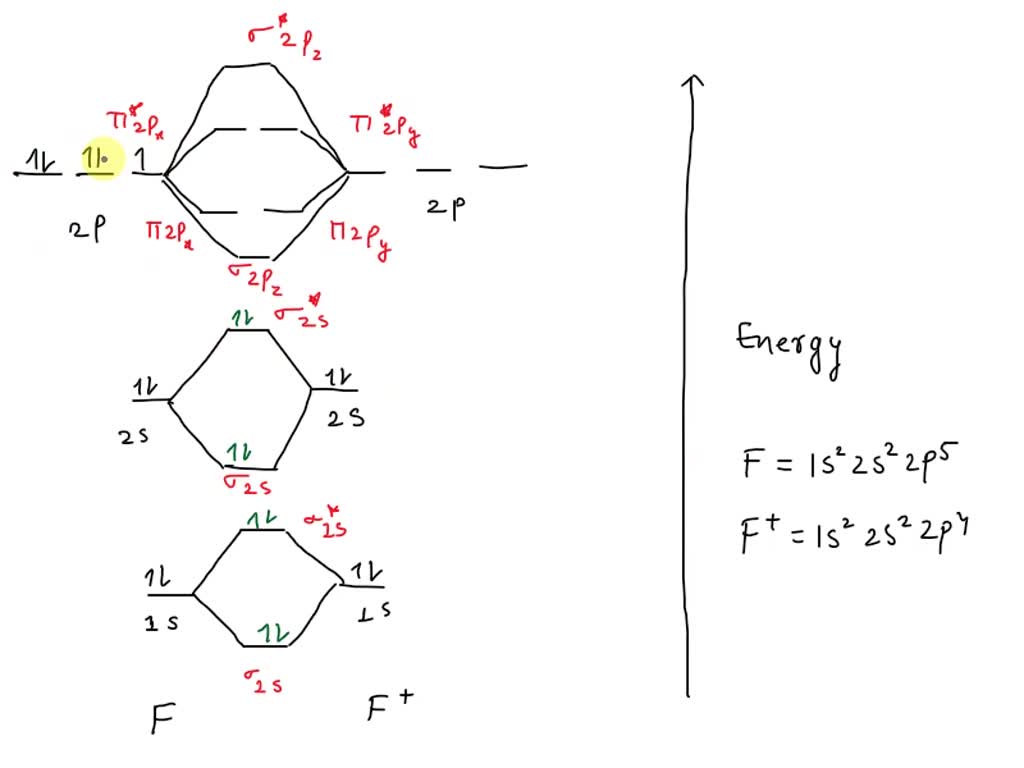
The Twist: Experimental Evidence
Here's where things get interesting! For years, simplified molecular orbital theory said F2 should be paramagnetic. Makes sense, right? Like its oxygen neighbor.
But, experiments disagreed. F2 stubbornly refused to be attracted to a magnet! What gives?
Turns out, the energy levels of the sigma and pi orbitals in F2 are flipped. Specifically, the sigma 2p orbital is lower in energy than the pi 2p orbitals.
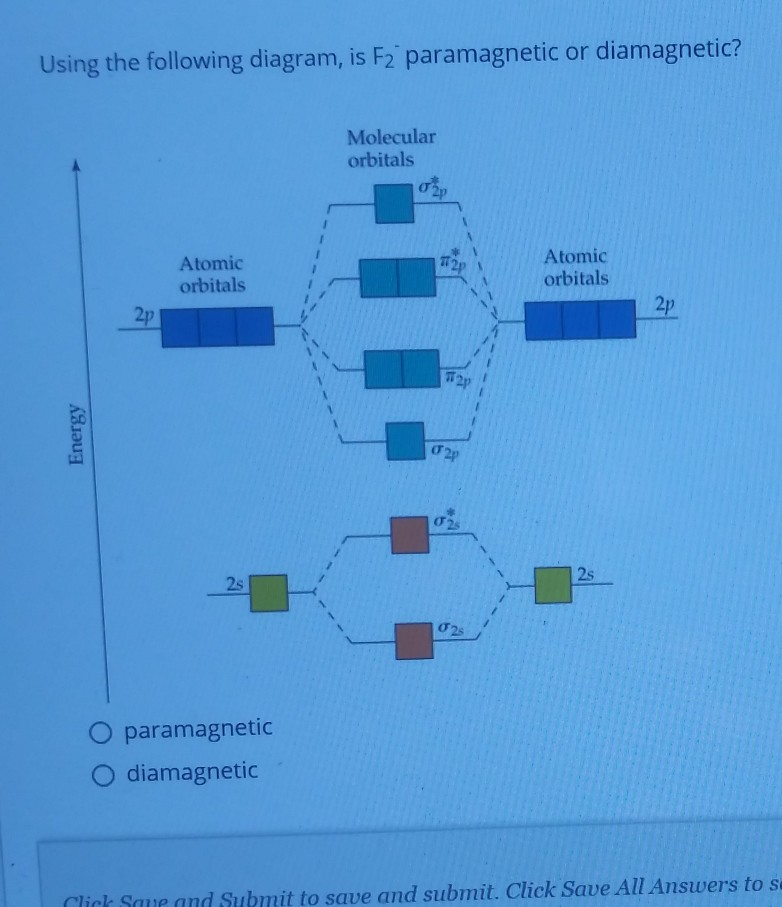
The reason? Electron-electron repulsion! Fluorine is a small atom with lots of electrons crammed into a small space. These electrons don't like being close to each other! This repulsion particularly affects the sigma 2p orbital, pushing it to a higher energy level. But the flipping of orbitals only happens in F2!
This swap of energy levels means all the electrons pair up in the molecular orbitals. No unpaired electrons are strutting their stuff.
This means F2 is diamagnetic! It's a bit of a surprise, honestly!
Why This Matters (Kind Of)
Okay, so knowing whether F2 is paramagnetic or diamagnetic isn't going to help you bake a cake. But, it highlights a crucial point: Chemistry is nuanced! Theoretical models are awesome, but experimental evidence is king (or queen!).
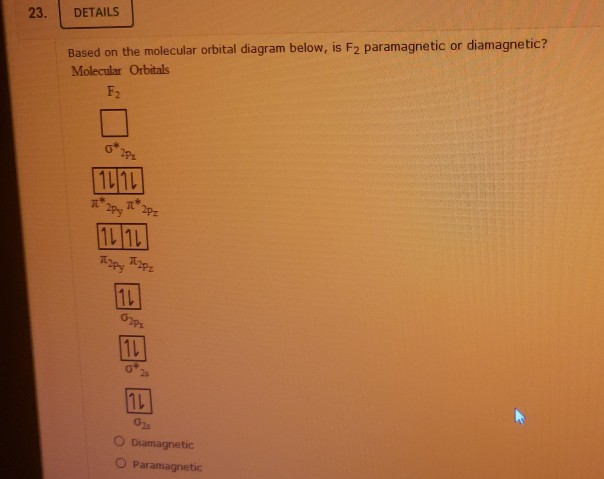
This also showcases the limitations of simplified models. While helpful for understanding basic concepts, they can sometimes lead to incorrect predictions. Molecular orbital theory, especially when used with computational chemistry, can take these effects into account and accurately predict the electronic structure and properties of molecules. Electron-electron repulsion is a real thing, and it can have surprising effects on molecular properties.
Think of it like this: You can have a general recipe for cookies, but sometimes you need to tweak it based on the specific ingredients you have. Same with chemistry!
The Fun Takeaway
F2 throws a wrench in our expectations. It’s a reminder that chemistry isn't always predictable. And that's what makes it so fascinating! The world is full of little surprises, even in the most seemingly simple molecules. Who knew that the seemingly basic question of whether F2 is paramagnetic or diamagnetic could lead to such an intriguing discussion about electron-electron repulsion and the limitations of simplified models?
So next time you encounter a seemingly simple scientific question, remember F2! Never underestimate the power of a good exception to the rule! Chemistry is fun because it's constantly challenging us and revealing new secrets.
