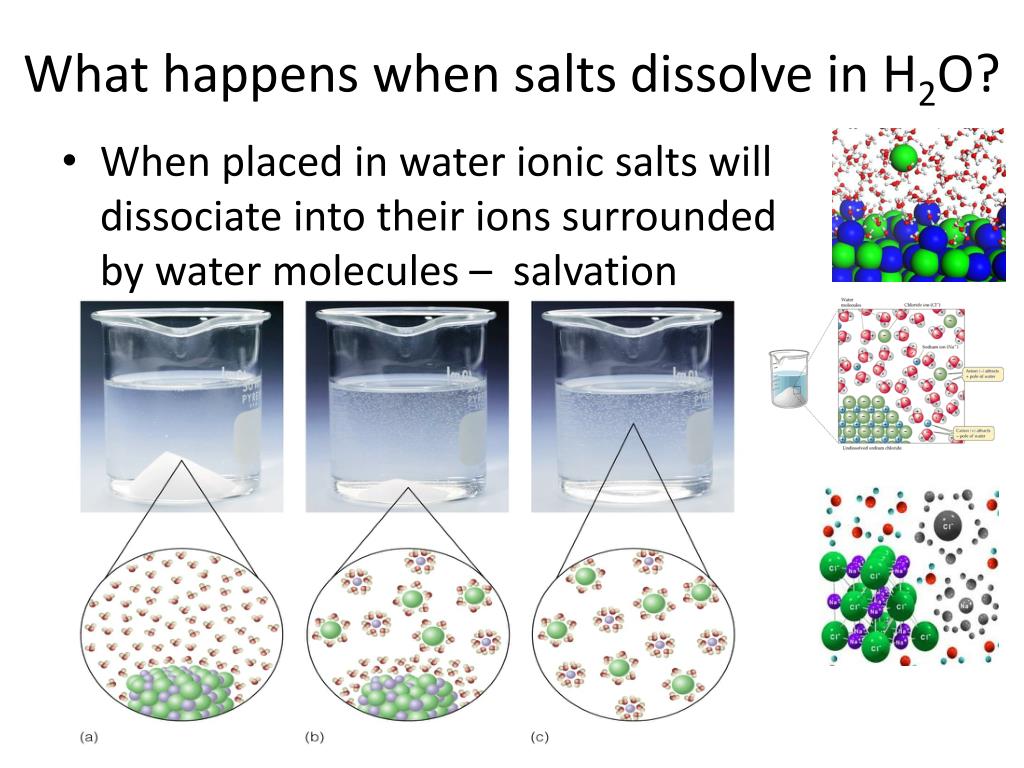
Is Dissolving Salt In Water A Physical Change

Ever watched salt seemingly vanish into water and wondered what magical transformation just occurred? It's not quite alchemy, but the simple act of dissolving salt in water is a fascinating example of a physical change, and understanding it opens doors to creativity and a deeper appreciation for the world around us. Forget complicated chemistry labs; this is science you can explore in your kitchen!
So, why should artists, hobbyists, or even just curious minds care? Because understanding this concept unlocks possibilities! For artists, it's the foundation for techniques like watercolor painting and dyeing fabrics. The way salt affects water's properties – like density and freezing point – influences how pigments spread, creating unique textures and effects. Hobbyists can use this knowledge to experiment with homemade solutions for cleaning, preserving, or even creating simple science experiments that are both educational and fun. And for the casual learner, grasping this principle is a stepping stone to understanding more complex chemical reactions. It's all about building a foundation of scientific literacy that makes the world a little less mysterious and a lot more engaging.
Let's look at some examples. In watercolor painting, artists use varying amounts of water to control the intensity of the color and the way it blends. Adding salt to a wet watercolor wash creates interesting textural effects as the salt crystals absorb the pigment-rich water, leaving behind lighter areas and a granulated appearance. Think of dramatic skies with textured clouds or the shimmering surface of water in a seascape. In tie-dye, dissolving salt in the dye bath helps the dye penetrate the fabric fibers more effectively, resulting in richer, more vibrant colors. This is especially useful for natural fibers like cotton and linen. Even something as simple as pickling vegetables relies on the principles of dissolving salt to draw moisture out of the food and preserve it. These are just a few examples; the possibilities are truly endless!
Must Read
Want to try some experiments at home? It's easy! First, gather some basic supplies: salt, water, a clear glass or jar, and a spoon. Start by adding a spoonful of salt to the water and stirring. Observe what happens. Does the salt disappear? Where does it go? Keep adding salt, one spoonful at a time, until no more salt will dissolve. This is called a saturated solution. You can also experiment with different temperatures of water. Does salt dissolve more readily in hot or cold water? Try creating a saturated salt solution and then slowly evaporating the water to see salt crystals form. Remember to always supervise children when conducting experiments involving water and other materials.
Why is this so enjoyable? Because it's tangible. You're not just reading about science in a textbook; you're experiencing it firsthand. You're observing the interactions between matter and energy, and you're using that knowledge to create something new or solve a problem. The simple act of dissolving salt in water is a reminder that science is all around us, waiting to be discovered and explored. It's a pathway to curiosity, creativity, and a deeper understanding of the amazing world we live in. So, go ahead, grab some salt and water, and start experimenting. You might be surprised at what you discover!