Is Copper A Solid Liquid Or Gas

Ever picked up a shiny penny or glanced at the copper pipes under your sink and wondered, "What exactly is copper, really?" It might seem like a simple question, but digging a little deeper into the nature of everyday materials like copper can be surprisingly fun and relevant to understanding the world around us. We often take for granted the states of matter – solid, liquid, or gas – but knowing why something behaves the way it does unlocks a whole new level of appreciation for everything from your smartphone to the weather outside.
So, let's get right to it: Is copper a solid, liquid, or gas? For most of us, most of the time, the answer is a resounding and obvious, solid! Think about it – you can hold a copper coin, it keeps its shape, and it’s certainly not sloshing around. This is because copper, like most metals, has a very stable atomic structure at typical room temperatures. Its atoms are tightly packed together and held in a fixed lattice by strong metallic bonds, which is the defining characteristic of a solid material. These strong bonds are what give copper its durability, excellent conductivity, and that satisfying weight.
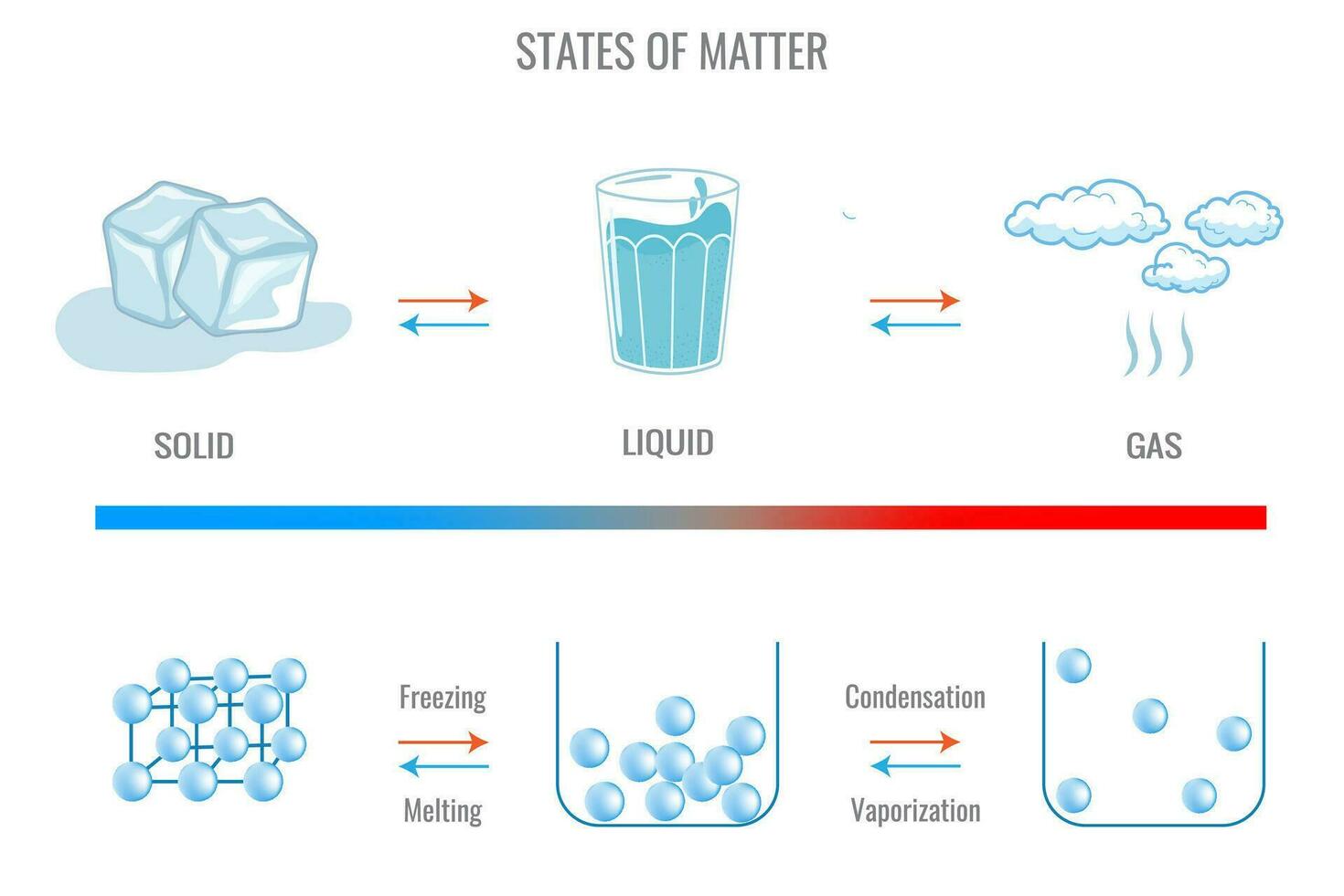
But here's where it gets interesting: while copper is definitely a solid in our daily experience, the truth is that any substance can exist in all three states of matter, given the right conditions. The "purpose and benefit" of understanding this lies in grasping the fundamental principles of physics and chemistry that govern our universe. It helps us understand how materials are formed, how they react to different environments, and how we can manipulate them for countless applications. For copper, transforming it into a liquid or gas requires some pretty extreme temperatures!
Must Read
To turn solid copper into a liquid, you'd need to heat it up to a scorching 1,085 degrees Celsius (1,984 degrees Fahrenheit). At this point, the atomic bonds weaken enough for the copper atoms to move freely past each other, creating molten copper – a shimmering, orange-hot liquid that's fascinating to watch (from a safe distance!). This liquid state is crucial in industries like metallurgy, where copper is melted down to be cast into new shapes, like wires, pipes, or intricate sculptures. Go even higher, to a blistering 2,562 degrees Celsius (4,643 degrees Fahrenheit), and copper will vaporize into a gas! You probably won't encounter gaseous copper in your kitchen, but understanding these phase changes is vital in fields like vacuum deposition or advanced material science.
In daily life, examples of copper as a solid are everywhere: electrical wiring carrying power to your home, plumbing pipes delivering water, and even in many beautiful works of art. In education, observing phase changes (even with water, which is far safer!) is a cornerstone of learning about matter and energy. When we learn about copper, we often discuss its use in electronics precisely because it's a stable solid with excellent electrical conductivity at room temperature.
So, how can you explore this yourself? While melting copper at home is definitely not recommended (it's extremely dangerous and requires specialized equipment!), you can appreciate the concept in simpler ways. Next time you see a copper wire, remember that its solid nature allows it to conduct electricity efficiently without losing its shape. Think about how blacksmiths heat and shape metals – they're bringing them close to their melting points to make them malleable. You can also explore videos online about metal casting or visit a science museum to see exhibits on materials and their properties. Just knowing that the solid copper in your pocket has the potential to become a glowing liquid or an invisible gas under the right conditions adds a touch of wonder to an otherwise ordinary object!