Is Ccl2h2 Polar Or Nonpolar

Ever wonder why some things mix well, like sugar in water, while others, like oil and water, resolutely refuse? It all boils down to something called polarity! And while that might sound like serious science, understanding it can be surprisingly fun and useful, especially when you start applying it to everyday stuff. Let's dive into the world of chemical compounds and explore whether CCl2H2, also known as dichloromethane, is polar or nonpolar.
So, why should you care if dichloromethane is polar or nonpolar? Well, for beginners, this is a fantastic introduction to understanding how molecules interact. It lays the groundwork for grasping concepts like solubility and chemical reactions. For families, think about cleaning products. Knowing about polarity helps you understand why some cleaners work better on grease (nonpolar) while others are better for dissolving water-based stains (polar). And for hobbyists, especially those into things like model building or crafting, understanding polarity can influence your choice of solvents and adhesives for different materials. Getting this right means your projects last longer and look better!
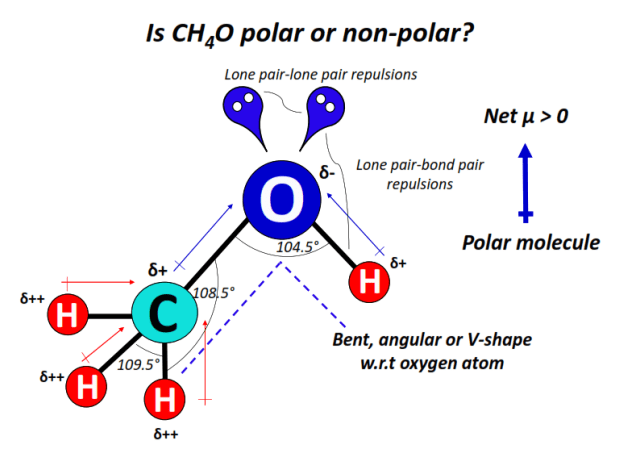
Think of polarity as a kind of electrical charge distribution within a molecule. If the charge is evenly spread, it's nonpolar. If there's an uneven distribution, creating a slightly positive and slightly negative end, it's polar. In the case of CCl2H2 (dichloromethane), we have two chlorine atoms and two hydrogen atoms attached to a central carbon atom. Chlorine is more electronegative than both carbon and hydrogen, meaning it pulls electrons towards itself more strongly. This creates a significant difference in electronegativity between the chlorine atoms and the rest of the molecule.
Must Read
Because of this uneven distribution of electron density, the chlorine atoms create a partial negative charge on their side of the molecule, while the hydrogen atoms and carbon experience a partial positive charge. This asymmetrical charge distribution makes CCl2H2 a polar molecule. It’s not super polar like water, but it definitely has a noticeable dipole moment (a measure of polarity).
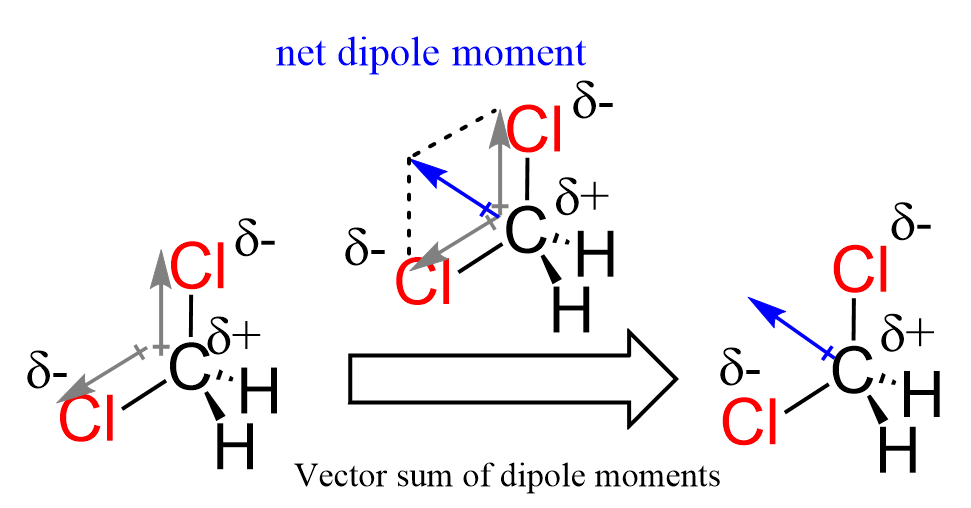
Variations on this theme are everywhere! For example, carbon tetrachloride (CCl4) has four chlorine atoms equally spaced around the carbon atom. Even though each C-Cl bond is polar, the symmetrical arrangement cancels out the individual bond dipoles, making CCl4 a nonpolar molecule. On the other hand, chloroform (CHCl3) has three chlorines and one hydrogen, making it polar like dichloromethane, but perhaps even more so.
Practical Tips for Getting Started:
- Visualize the Molecule: Draw the Lewis structure of CCl2H2. This helps you see the arrangement of atoms.
- Consider Electronegativity: Remember that chlorine is much more electronegative than hydrogen.
- Think About Symmetry: Is the molecule symmetrical? If not, it's likely polar.
- Use Online Resources: There are tons of websites and videos that explain polarity. Search for "dichloromethane polarity" to find them.
Understanding the polarity of molecules like CCl2H2 opens a door to a fascinating world. It’s not just about memorizing facts; it’s about developing an intuition for how molecules interact and why things behave the way they do. Whether you're curious about chemistry, trying to find the right cleaning product, or tackling a crafting project, a little knowledge about polarity can go a long way. So, have fun exploring the world of molecules – it's more interesting than you might think!