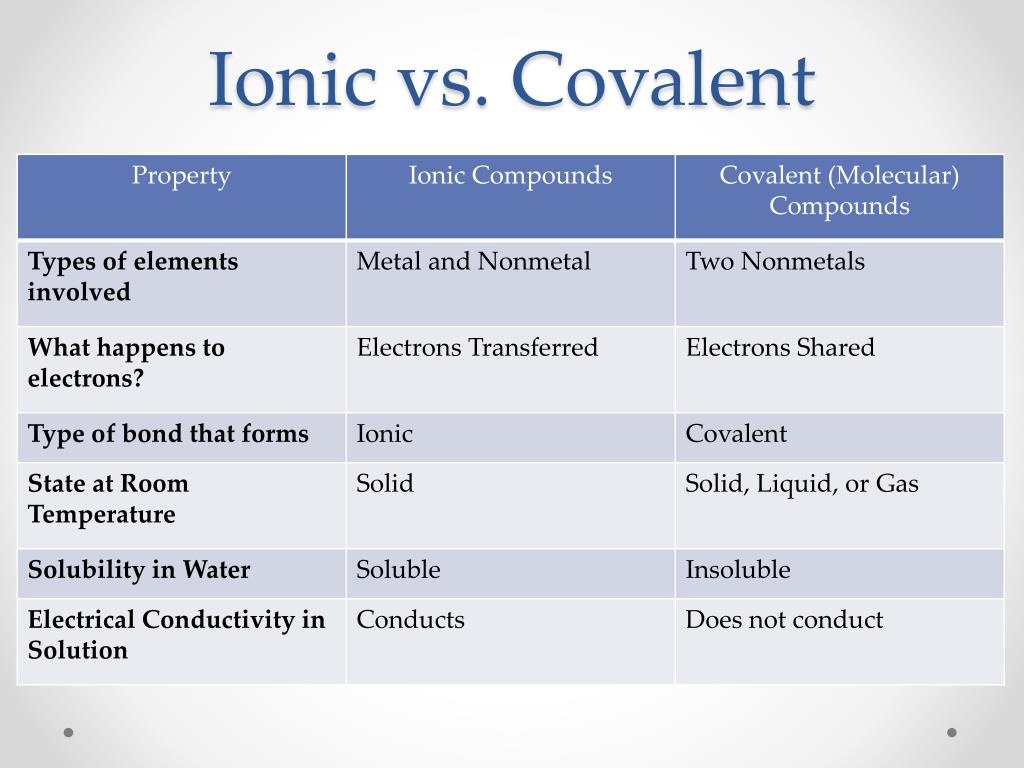
Is C6h12o6 Ionic Or Covalent

Ever heard of C6H12O6? Probably not by that name! But what if I said "glucose" or "fructose"? Suddenly, it rings a bell, right? This little molecule, the simple sugar so fundamental to life, isn't just about biology and chemistry textbooks. It's also a fascinating example of how things stick together at the atomic level, and understanding it can unlock some surprising creative insights, even if you’re not a scientist!
So, is C6H12O6 ionic or covalent? The answer is definitively covalent. Why should artists, hobbyists, and casual learners care? Well, grasping the concept of covalent bonds helps you appreciate the inherent stability and structure of many things around you. Think about the materials you use every day. Paints, glues, fabrics – a vast majority are held together by covalent bonds. Knowing this gives you a deeper understanding of their properties. For example, understanding how molecules interact helps an artist choose the right mediums for layering and blending, knowing which paints will adhere well and which might crack or peel over time.
Imagine you're creating a watercolor painting. The water molecules (H2O, also covalent!) interact with the paper fibers, allowing the pigments to spread and blend. Knowing that these interactions are based on sharing electrons, rather than transferring them as in ionic bonds, can make you more mindful of the delicate balance needed for a successful wash. Or perhaps you're a baker, meticulously crafting a cake. The sugar (C6H12O6!) caramelizes due to heat, breaking and reforming covalent bonds, leading to those delicious flavors and textures. Appreciating this chemical dance elevates your understanding and allows for more informed experimentation in the kitchen.
Must Read
Here's an example: think of the difference between table salt (NaCl, ionic) and a sugar cube (C6H12O6, covalent). Salt crystals are hard and brittle, easily dissolving in water into separate ions. Sugar, while crystalline, is generally more pliable and dissolves differently, its molecules staying largely intact. This is a direct result of the different types of bonds holding them together. Explore different art styles! Covalent bonds make things stay firmly put. This can apply to the detailed and planned art style, to demonstrate stability. In contrast, ionic bonds can display an art style that is more sporadic and inconsistent.
Want to try exploring this at home? A simple experiment could be comparing how quickly salt and sugar dissolve in water, paying attention to the visual differences in the process. Or, for a more creative approach, try making two different types of glues: one using sugar and water (a simple syrup glue) and another using salt and water. Compare their adhesive properties. Observe how different the structures are, and reflect on the type of bonds present within the chemical component.
Ultimately, understanding whether C6H12O6 is ionic or covalent isn’t just about memorizing facts. It's about opening your eyes to the invisible world of molecules and appreciating the fundamental forces that shape our reality. It's about fueling your curiosity and finding new ways to connect science and creativity. Plus, it’s pretty cool to know you’re pondering the very building blocks of life every time you enjoy a sweet treat!