Is C2 Paramagnetic Or Diamagnetic

Okay, let's talk about molecules! Specifically, the molecule C2, which is just two carbon atoms bonded together. And we're going to figure out if it's paramagnetic or diamagnetic. Now, before your eyes glaze over, trust me, this is actually kind of cool, and I'll explain why you should even care in the first place.
Think of it like this: Imagine you're at a party and some people are naturally drawn to each other (paramagnetic), while others prefer to keep their distance and not interact much with the crowd (diamagnetic). That’s a very simplified analogy, of course, but it gives you the general idea!
What's the Big Deal Anyway? Why Should I Care?
Good question! Why should you care whether a molecule is paramagnetic or diamagnetic? Well, it boils down to how it interacts with magnetic fields. You know, like the ones that make your fridge magnets stick? Understanding this can tell us a lot about a molecule's structure, its stability, and how it might react with other molecules. In fields like materials science and chemistry, this stuff is incredibly important for designing new materials, drugs, and technologies!
Must Read
Imagine you're designing a new super-strong material for, say, airplane wings. Knowing if the molecules that make up that material are paramagnetic or diamagnetic can help you predict how it will behave under stress and in different environments. Or, if you're developing a new drug, you want to know how it will interact with the body, right? Understanding the magnetic properties of the drug molecule can give you clues about its behavior.
Paramagnetic vs. Diamagnetic: The Short Version
So, what's the difference? It all comes down to electrons. Remember those little guys buzzing around the nucleus of an atom? Electrons like to hang out in pairs, like comfy socks. When all the electrons in a molecule are paired up, that molecule is diamagnetic. It's like a group of people at our party where everyone has a partner and no one is really looking for attention outside their pair. Diamagnetic substances are slightly repelled by a magnetic field.

However, if a molecule has unpaired electrons – lonely socks, if you will – it's paramagnetic. These unpaired electrons act like tiny magnets themselves. Paramagnetic substances are attracted to a magnetic field. They're like people at the party looking for someone to pair up with, actively seeking attention from others around them.
So, is C2 Paramagnetic or Diamagnetic? The Plot Thickens...
This is where things get a little tricky. You might think, "Okay, two carbons, each with four valence electrons... they should pair up nicely!" And for a long time, that's what scientists thought too! But experiments showed something different.
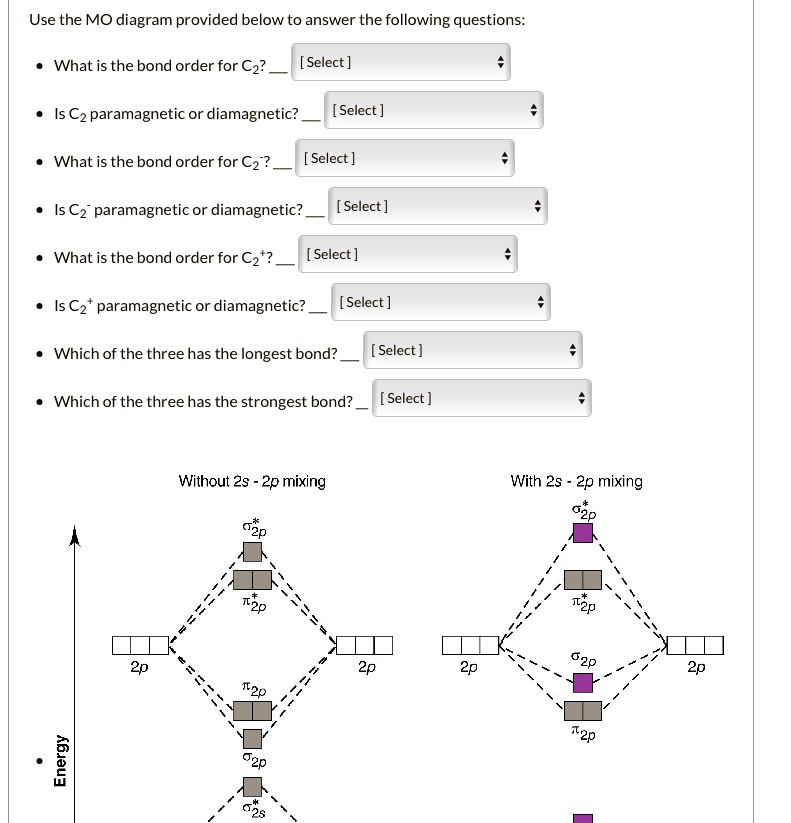
The story of C2 is actually a great example of how science works. Early theories suggested that C2 would have a quadruple bond with all electrons paired. But experimental data proved that C2 is, in fact, paramagnetic in its ground state!
That's because the way the electrons are arranged in C2 is a bit unusual. Quantum mechanics tells us that the electrons fill molecular orbitals, which are like energy levels for the whole molecule, not just individual atoms. In the case of C2, the two highest occupied molecular orbitals are both partially filled, each containing one unpaired electron. This leads to C2 having two unpaired electrons, making it a paramagnetic molecule.

Think of it like this: instead of two people directly holding hands, they're each holding onto a separate rope, and both ropes are tied to a central point. It's a connection, but it's a bit more... loose. This "looseness" allows the electrons to remain unpaired.
Why the Surprise?
The fact that C2 is paramagnetic was a big surprise because it challenges our intuition about how bonds typically form. We often think of bonds as being formed by paired electrons, but C2 shows us that things can be more complex. This unexpected finding highlighted the limitations of simple bonding models and pushed scientists to develop more sophisticated theories.
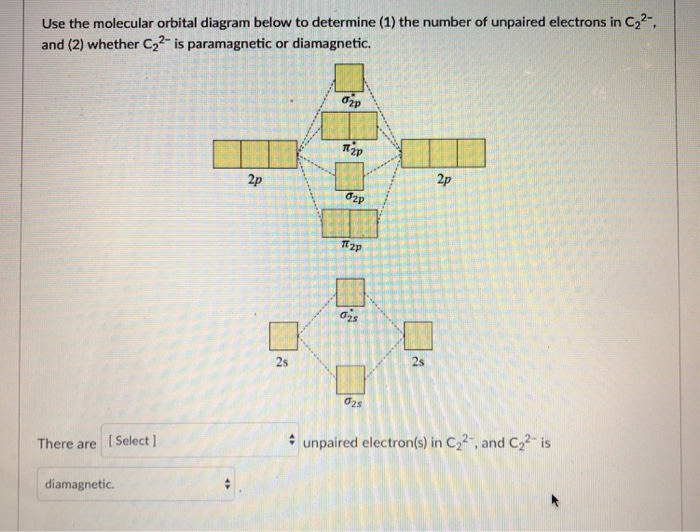
Essentially, understanding the electronic structure of C2 required looking at the problem from a more advanced quantum mechanical perspective.
The Takeaway
So, there you have it! C2 is paramagnetic. It might seem like a tiny detail in the grand scheme of things, but it illustrates some fundamental principles of chemistry and physics. It highlights the importance of experimental evidence, the power of quantum mechanics, and the fact that even seemingly simple molecules can have surprising and fascinating properties.
Next time you see a magnet sticking to your fridge, remember C2 and its unpaired electrons. It's a reminder that the world around us is full of tiny, interacting forces, and that even the smallest details can have a big impact.
