Is Aluminum A Pure Substance Or Mixture

Ever wonder what everyday stuff is really made of? It's more fascinating than you think! Understanding whether something is a pure substance or a mixture is like unlocking a secret code to the world around us. Today, we're diving into the curious case of aluminum: is it a pure substance or a mixture?
Why should you care? Well, for beginners, it's a fantastic introduction to the basics of chemistry. For families, it can spark interesting conversations and even lead to fun science experiments at home. And for hobbyists, especially those who tinker with metalworking or DIY projects, knowing the composition of your materials is essential for success.
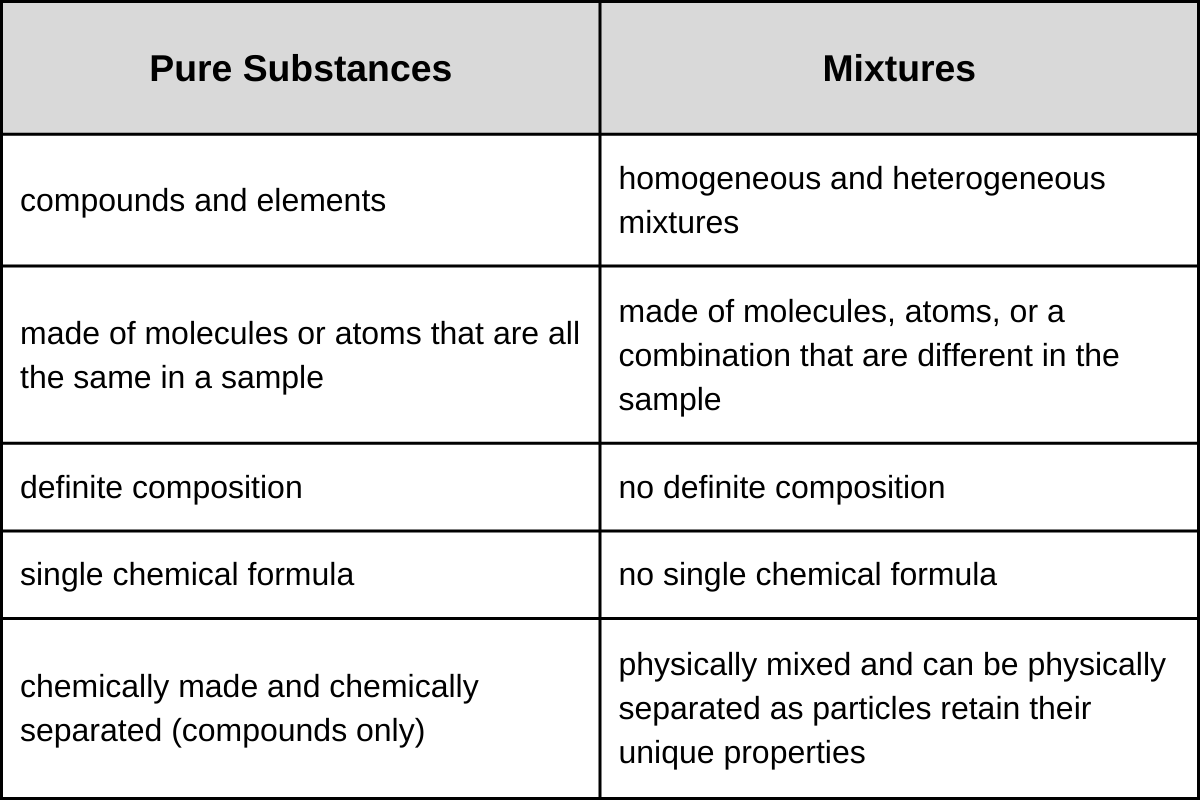
So, what's the verdict on aluminum? The answer is: Aluminum, in its elemental form, is a pure substance. A pure substance is made up of only one type of atom or molecule. In aluminum's case, it's all aluminum atoms (Al). Think of it like this: if you could zoom in super close, you'd only see aluminum atoms packed together.
Must Read
But hold on! Things get a little trickier because we rarely encounter pure, pure aluminum in everyday life. Most aluminum products are actually alloys. An alloy is a mixture of metals designed to enhance specific properties. For example, the aluminum used in airplanes is often an alloy containing small amounts of copper, magnesium, or silicon. These additions make the aluminum stronger and more resistant to corrosion.
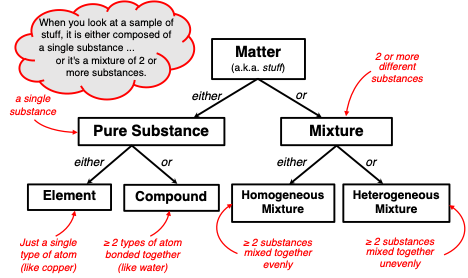
So, while elemental aluminum is a pure substance, the aluminum foil in your kitchen or the aluminum can you're holding is likely an alloy – a carefully crafted mixture. This difference is important. Pure aluminum is soft and easily dented, while aluminum alloys are much more durable.
Here are a few examples to illustrate the difference:

- Pure Aluminum: Used in specialized applications where its conductivity and corrosion resistance are paramount. Think of high-purity aluminum wires in electronics.
- Aluminum Alloy: Used in construction, transportation (cars, airplanes), packaging (cans), and cookware because of its strength and durability.
Want to get started exploring pure substances and mixtures? Here are some simple tips:
- Look at Labels: Start reading the labels on everyday products. See if you can identify pure substances or mixtures.
- Simple Experiments: Try dissolving sugar in water. Is the result a pure substance or a mixture? What happens if you evaporate the water?
- Explore Online: There are tons of free resources online that explain these concepts in more detail, often with interactive simulations.
Understanding the difference between pure substances and mixtures opens up a whole new way of looking at the world. It's like having a superpower that allows you to understand the composition and behavior of everything around you. So, embrace the curiosity, experiment, and enjoy the journey of exploring the fascinating world of chemistry!