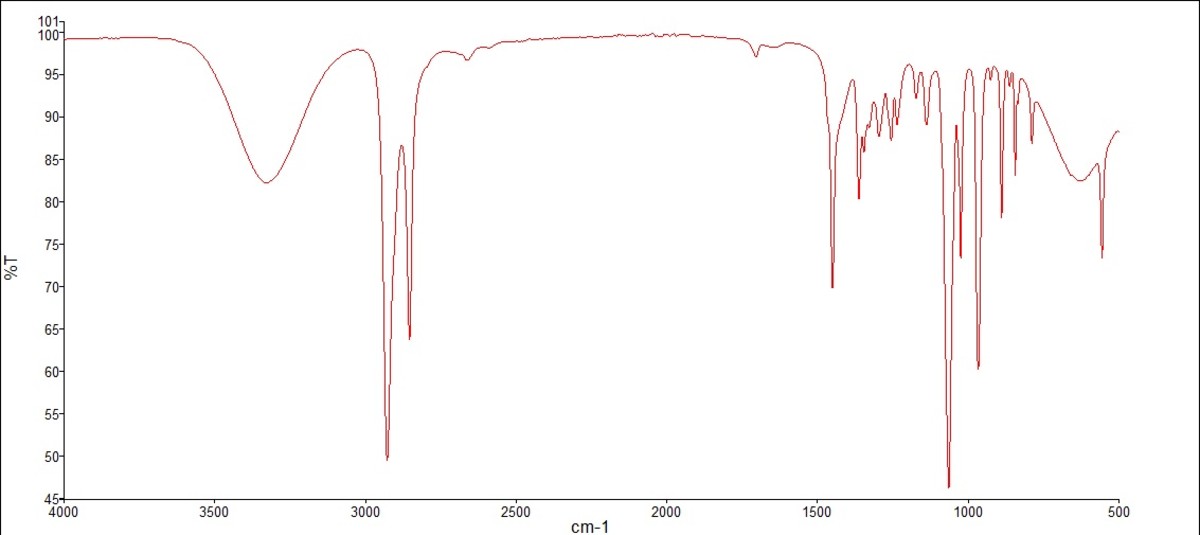
Ir Spectrum For Cyclohexanone
Okay, so IR Spectroscopy for cyclohexanone. Sounds intimidating, right? Like something a mad scientist would yell while surrounded by bubbling beakers. But trust me, it's not rocket surgery (unless you ARE a rocket surgeon, in which case, kudos!). Think of it more like… decoding the secret language of molecules using light. Bear with me!
We're talking about cyclohexanone, which, let's be honest, sounds like a villain in a B-movie. But it's actually a perfectly respectable, ring-shaped molecule. And it's got a secret: It vibrates.
Everything vibrates, really. You, me, your annoying neighbor's leaf blower at 7 AM. But molecules vibrate at specific frequencies, like tiny little tuning forks. And the beauty of IR spectroscopy is that it can detect these vibrations. It's like listening to the molecular orchestra and identifying which instruments are playing.
Must Read
Imagine shining a special flashlight – an infrared light – at your cyclohexanone sample. Some of that light will pass right through, no problem. But some of it will get absorbed. Why? Because the molecule's vibrating at the exact same frequency as the light. It's like when you push a kid on a swing at just the right rhythm, and they go higher and higher. The molecule absorbs the energy and vibrates even more!
The Key Player: The Carbonyl Group (C=O)
Now, the really important part of cyclohexanone is its carbonyl group (C=O). That's a carbon atom double-bonded to an oxygen atom. Think of it as the "diva" of the molecule. It's the one that really steals the show in the IR spectrum. Why? Because that C=O bond vibrates very strongly, and its absorption band is super obvious, like a giant neon sign screaming, "I'M HERE!".
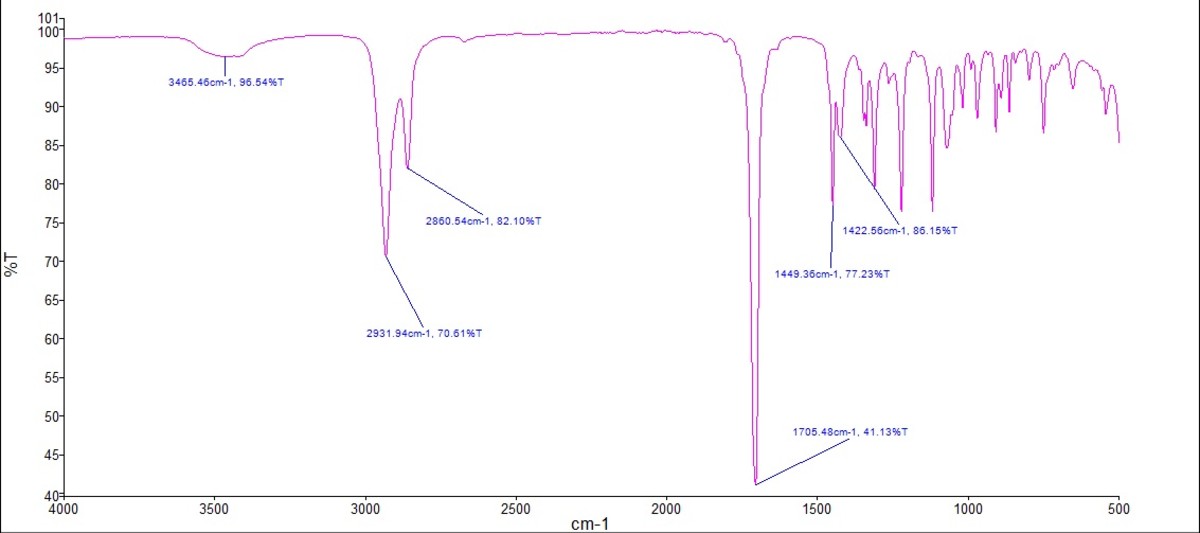
This C=O stretch usually shows up in the IR spectrum somewhere around 1715 cm-1 (wavenumbers, a fancy way of measuring frequency). That's a pretty distinctive number. It's like having a molecular fingerprint. If you see a strong peak in that area, BOOM! You've likely got a carbonyl group. Congrats, you are now a part-time molecular detective.
Think of it like this: you walk into a room and smell freshly baked cookies. You immediately think, "Aha! Cookies!". That 1715 cm-1 peak is the "freshly baked cookies" smell of the carbonyl group.
Other Vibrations, Other Clues
Of course, cyclohexanone has other vibrations too. It has C-H stretches (around 2850-3000 cm-1), which are like the background music to the C=O diva's performance. They're there, but not quite as attention-grabbing. These are basically the molecule's polite way of saying, "Yeah, I'm made of carbon and hydrogen too."
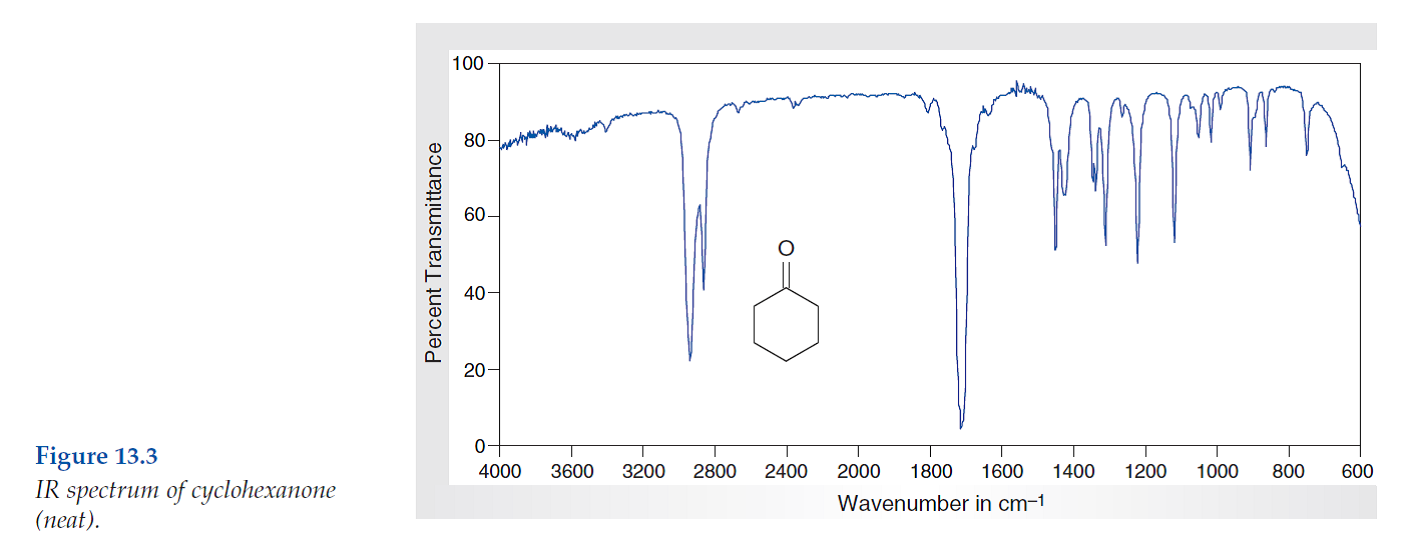
You might also see some C-C stretches and bends in the fingerprint region (below 1500 cm-1), which are, frankly, a bit of a mess to interpret. It's like trying to decipher your toddler's abstract painting. You know something's going on, but good luck figuring out exactly what.
Interpreting the Spectrum: It's Not Always Black and White
Interpreting an IR spectrum isn't always as easy as spotting the C=O peak and calling it a day. Sometimes, other things can affect the position and shape of the peaks. For example, if the carbonyl group is part of a larger, more complex molecule, its peak might shift slightly. It's like the cookie smell being fainter if the oven is farther away.
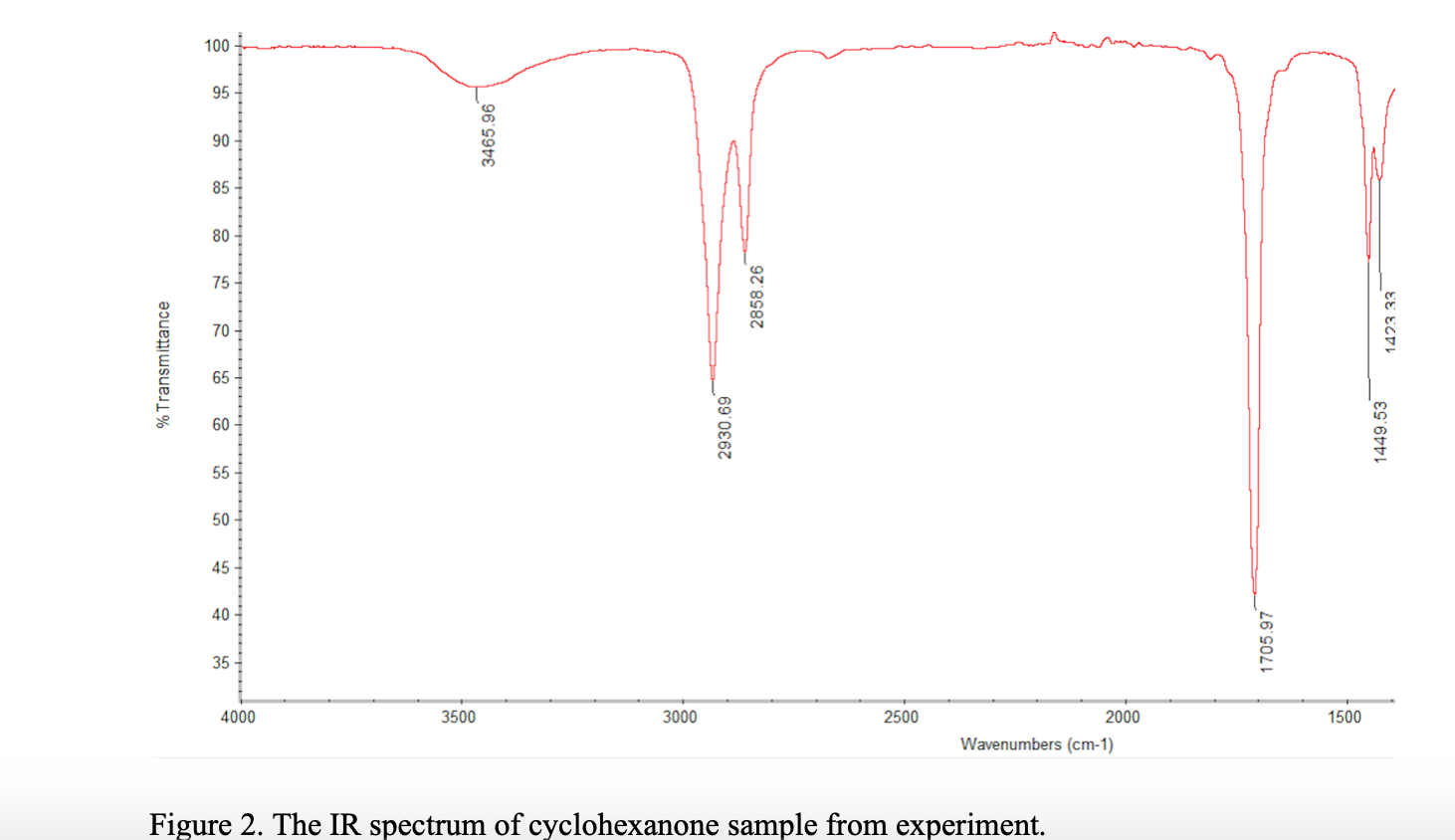
But generally, if you see that strong peak around 1715 cm-1, and you know you're dealing with a ketone, you're probably looking at cyclohexanone (or something very similar).
So, to recap:
- IR spectroscopy is like shining a special light on molecules to see how they vibrate.
- Cyclohexanone has a super important carbonyl group (C=O).
- The C=O stretch shows up around 1715 cm-1 and is like the molecule's fingerprint.
- Other vibrations exist, but the C=O is the main event.
Next time you hear about IR spectroscopy, don't panic! Just remember the vibrating molecules, the diva carbonyl group, and the freshly baked cookie smell. You've got this!