Ir Spectra For Benzoic Acid

Ever baked a cake and wondered what makes it, well, cake? Or perhaps pondered the secret to that amazing aroma wafting from your grandma's kitchen? A big part of the answer, surprisingly, can be found by looking at something called an IR spectrum. And today, we're going to explore this fascinating tool through the lens of a familiar friend: benzoic acid.
Benzoic acid? Sounds scary, right? Like something out of a science fiction movie. But trust me, it's more 'deliciously preservative' than 'ominously alien'. In fact, it's a naturally occurring compound found in many fruits, like cranberries and plums. It's even used as a preservative in some foods, keeping things fresh and preventing unwelcome guests (bacteria and mold, mostly) from crashing the party.
So, what does this have to do with those IR spectra we mentioned earlier? Well, imagine benzoic acid as a tiny orchestra, each atom vibrating and wiggling in its own special way. An IR spectrum is like a recording of that orchestra, capturing the unique vibrational 'song' of the molecule. This song is far more interesting than a simple note. It is more like a complex musical arrangement and contains a wealth of structural information!
Must Read
The Infrared Spotlight: Exposing Benzoic Acid's Secrets
Now, let's crank up the 'IR spectrometer', our high-tech microphone, and listen to benzoic acid's symphony. This machine shines infrared light – the same type of heat radiation that keeps you warm on a cold day – through a sample of benzoic acid. As the light passes through, some of it is absorbed by the molecules, causing them to vibrate even more intensely. The specific wavelengths of light that are absorbed reveal which types of bonds are present in the molecule, like clues in a chemical detective story.
Think of it like this: imagine you're tuning a guitar. Each string has a unique natural frequency, and when you pluck it, it vibrates at that frequency. Similarly, each type of chemical bond in benzoic acid – like the bond between carbon and oxygen in the carboxyl group (-COOH) – has its own preferred vibrational frequency. This frequency is measured in the IR spectrum and shows up as a peak, or a dip, in the graph.

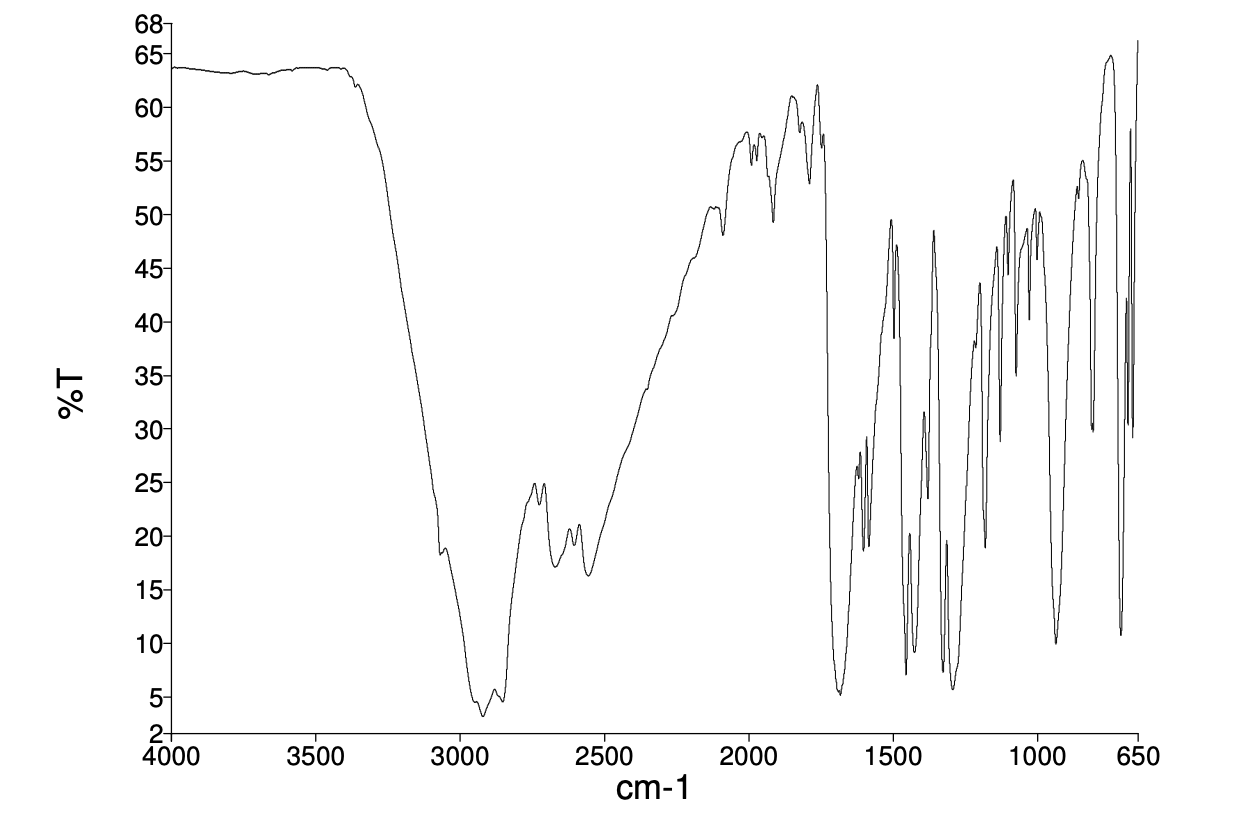
So, what does benzoic acid's IR spectrum tell us? Well, a big, broad peak usually appears around 2500-3300 cm-1. This is the telltale sign of the O-H bond in the carboxyl group. It's like benzoic acid shouting, "Hey, I've got an acid part here!" The broadness comes from the fact that these O-H bonds are often involved in hydrogen bonding, meaning they're interacting with other benzoic acid molecules like tiny, clingy magnets.
Then, there's another sharp peak around 1700 cm-1, the signature of the C=O bond, or carbonyl group. This one is like the bass line of benzoic acid's song, a clear and strong indication of the presence of a carbonyl group, which is characteristic of carboxylic acids.
![Benzoic Acid Ir 5 Amino 2 Hydroxy Benzoic Acid Optional[FTIR]](https://chemistrytalk.org/wp-content/uploads/2023/03/benzioc-acid-spectrum-1-1024x383-1.png)
And let's not forget the aromatic ring! Benzoic acid is, after all, "aromatic", meaning it contains that special ring structure that gives many compounds their characteristic smells and properties. These appear as multiple sharp peaks around 1450-1600 cm-1. This is like the complex melody of the orchestra, a collection of signals that show the presence of an aromatic ring.
Beyond the Peaks: The Heart of Chemistry
What's truly remarkable is that this seemingly abstract data – a graph with peaks and dips – allows chemists to identify and understand the structure of molecules. It's like having a molecular fingerprint, a unique signature that allows us to distinguish benzoic acid from other similar molecules.
But even more important is that it gives us insight into how the molecule behaves. It helps us understand how it interacts with other molecules, how it might react in certain conditions, and how it ultimately contributes to the world around us. Understanding IR spectra has far reaching applications in numerous fields, from pharmaceutical science and drug discovery, to understanding the composition of polymers, and identifying the components of unknown substances.
So, the next time you see a cranberry sauce, remember benzoic acid, our silent guardian of freshness. And remember the IR spectrum, the invisible orchestra that reveals the inner workings of this seemingly simple molecule. It's a reminder that even the smallest things, when viewed through the right lens, can hold incredible secrets and beauty.
"Chemistry is not about test tubes and solutions, but about understanding the world at its most fundamental level." – An anonymous chemist, probably.
Maybe, just maybe, you'll even start hearing the faint music of molecules yourself. Just kidding (mostly)! But hopefully, you'll see a little bit more magic in the ordinary, thanks to the fascinating world of IR spectra and our friend, benzoic acid.

