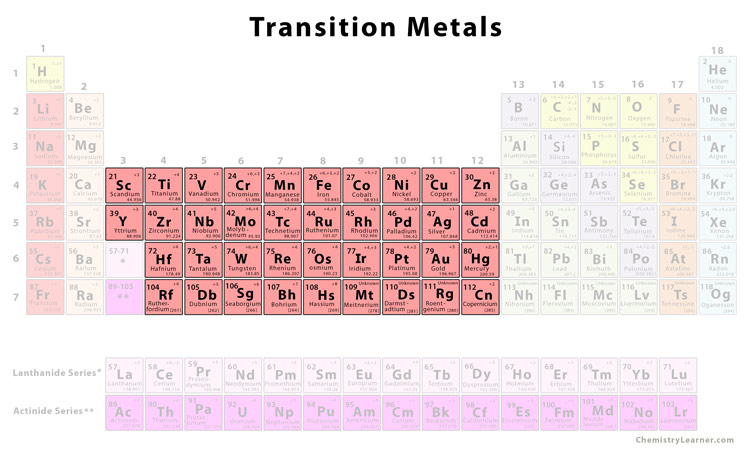
In What Section Would Transition Metals Be Found

Okay, chemistry class might feel like a distant memory, or maybe it's something you're currently wrestling with. Either way, let's talk about a fascinating group of elements: the transition metals. They're the workhorses of the periodic table, the chameleons of the element world, and knowing where to find them is key to understanding their unique properties.
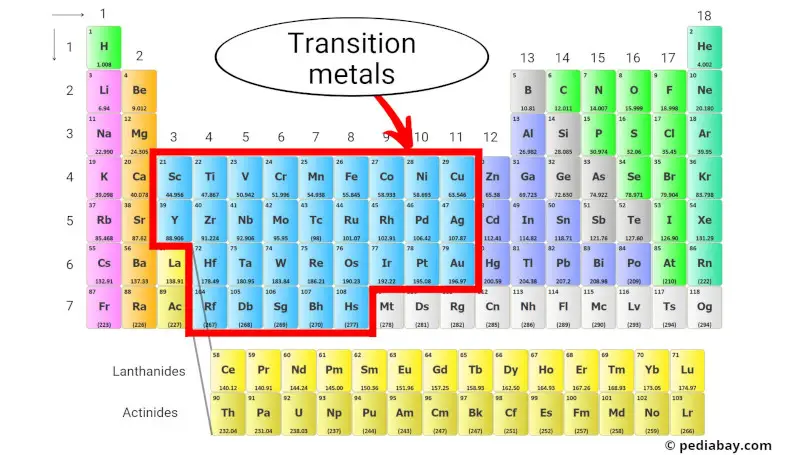
So, where do these metallic marvels hang out? Think of the periodic table like a city, and transition metals live in a specific neighborhood. They reside in the d-block, which is smack-dab in the middle of the periodic table. Forget the far left (alkali metals) and the far right (halogens and noble gases); we're talking center stage here!
The Periodic Table Neighborhood: A Guided Tour
Imagine the periodic table as a map. The alkali and alkaline earth metals (Groups 1 and 2) are like the trendy downtown area, always reacting and super active. Then comes a less densely populated region (Groups 3-12): this is where the transition metals live. Then you have the main group elements (Groups 13-18), the old, established neighborhood with familiar faces, like carbon and oxygen. Finally, the lanthanides and actinides form a quiet suburb at the bottom.
Must Read
The transition metals occupy Groups 3 through 12. You can’t miss them! They form a bridge between the highly reactive alkali and alkaline earth metals on the left and the less reactive main group elements on the right.
Think of it like this: the s-block is the opening act, the p-block is the headliner, and the d-block transition metals are the versatile band in the middle, able to play a little of everything and hold the whole show together.
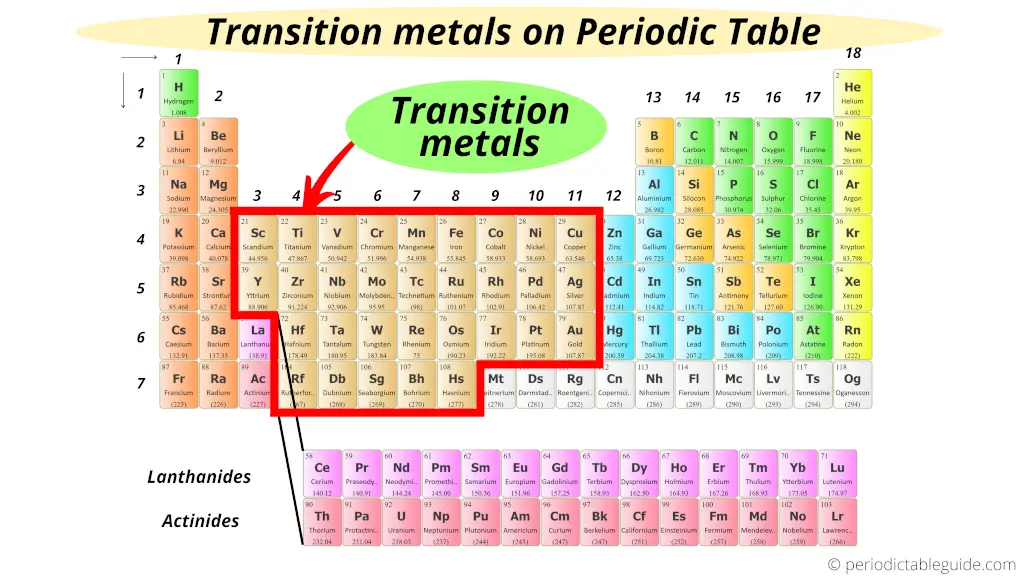
Why the Middle Matters: Unique Properties
Being in the middle isn't just about location; it's about characteristics! Transition metals have several distinguishing traits:
- Variable Oxidation States: They can form ions with multiple positive charges. This gives them a rainbow of possibilities when forming compounds, which explains why they are used as catalysts.
- Colored Compounds: Ever wonder why some gemstones are vibrantly colored? Often, it's thanks to transition metals! Think of the blue in sapphire (titanium and iron) or the green in emerald (chromium).
- Good Catalysts: Many transition metals are fantastic catalysts, speeding up chemical reactions. This is why they are so important in industry.
- High Melting and Boiling Points: These are tough metals! This makes them useful in structures.
For example, iron (Fe), a quintessential transition metal, is the backbone of our infrastructure, found in steel used in bridges and buildings. Copper (Cu), another transition metal, is essential for electrical wiring because of its excellent conductivity. Gold (Au) and silver (Ag) are used in jewelry and electronics. These are just a few examples of how we use transition metals everyday.
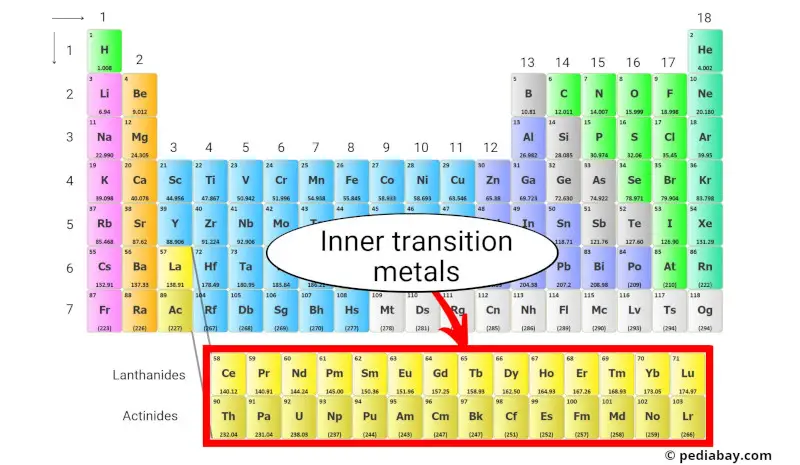
Mnemonic Fun: Remembering the Block
Need a trick to remember where to find them? How about this: "Dance in the D-block!" Silly, but memorable. Also, remember that the "d" comes after "s" and before "p" in the alphabet, just like the location on the periodic table.
Or, you could think of the names themselves: "Transition" implies a bridge, a connection. These metals transition between the s-block and p-block elements.
Beyond the Classroom: Transition Metals in Real Life
You might not realize it, but transition metals are everywhere! From the stainless steel in your kitchen sink to the pigments in your favorite paints, these elements play a crucial role in modern life. Even the hemoglobin in your blood, which carries oxygen, contains iron, a vital transition metal.
Next time you see a colorful piece of jewelry, admire a towering skyscraper, or even just breathe, remember the transition metals working behind the scenes. They're not just elements on a chart; they're essential building blocks of our world.
A Little Reflection
Understanding the periodic table isn't just about memorizing facts; it's about appreciating the fundamental building blocks of the universe. The transition metals, with their unique properties and versatile applications, remind us that even seemingly abstract concepts have tangible and significant impacts on our daily lives. So, next time you look at something made of metal, take a moment to appreciate the transition metals that made it all possible. They are found right in the middle, doing all the heavy lifting in style.