Hydrogen Is Metal Nonmetal Or Metalloids
Okay, so you’re at a party, right? And there’s always that one person. You know, the one who just… doesn’t quite fit in any group. They’re hanging out near the snacks, maybe trying to join the sports convo, but then they’re suddenly talking about art with the artists. That, my friends, is hydrogen in the periodic table. It’s the ultimate social chameleon.
The Identity Crisis of an Element
We're talking about hydrogen. Is it a metal? A nonmetal? Or maybe one of those in-between metalloid types? The answer, as frustrating as it is, is… it depends!
Think of metals. Shiny, conduct electricity well, love to give away electrons. Nonmetals are usually dull, bad conductors, and love to steal electrons. Metalloids? They’re like the undecided voters of the element world – sometimes they act like metals, sometimes like nonmetals.
Must Read
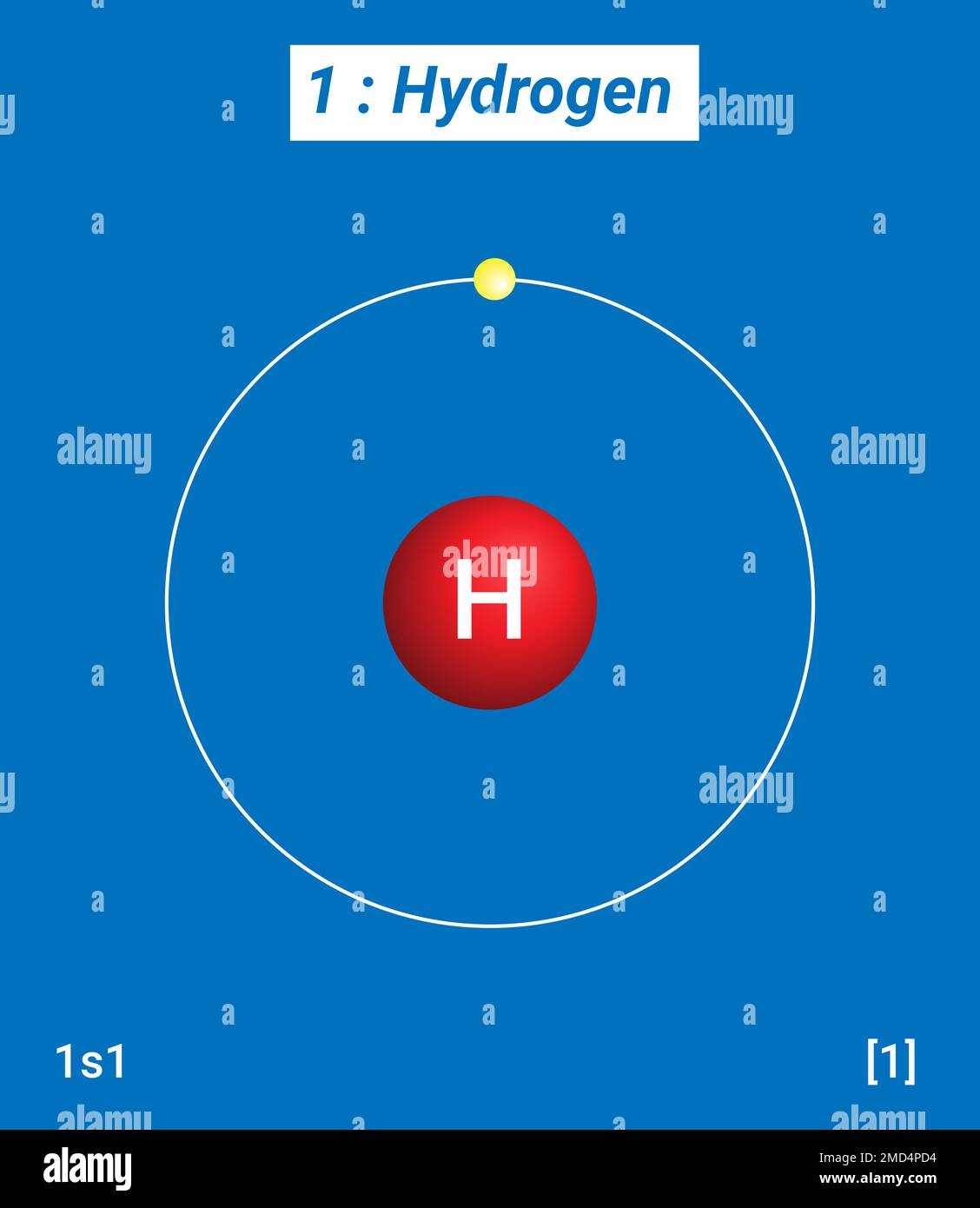
Hydrogen, bless its little single-proton heart, is none of those... and also kind of all of them! At room temperature and pressure, it’s a gas, making it a nonmetal by default. You wouldn't try to hammer it into a sword or string it up to power your phone. It is light and very reactive. So, so far, it looks like hydrogen is a nonmetal.
The Metallic Side of Hydrogen
But here’s where things get wild. Compress hydrogen enough – like, really enough, think pressures found deep inside Jupiter – and something magical happens. The hydrogen atoms get squeezed so close together that their electrons start behaving like they do in metals. Suddenly, you've got metallic hydrogen! Boom! Mind blown.

Imagine taking a balloon full of hydrogen and squeezing it with the force of, say, a million elephants standing on it. That's the kind of pressure we're talking about. Then, BAM! Superconducting, electrically conductive, shiny, potentially revolutionary metallic hydrogen!
The catch? It's incredibly hard to create and maintain. We're talking about lab conditions that make even the most seasoned scientist sweat a little.

But Wait, There's More! (The Nonmetal Part Strikes Back)
Even without the elephant-squeezing, hydrogen is still kind of weird. Its electron configuration (the way its electrons are arranged) is similar to alkali metals like lithium and sodium. Like them, it only needs to lose one electron to become stable. That's why it often hangs out in Group 1 of the periodic table. But unlike those metals, hydrogen can also gain an electron to become stable, just like a halogen nonmetal such as fluorine or chlorine!
It is this ability to either give or take electrons depending on the element that it bonds with that really makes hydrogen a chameleon element.

So, it can lose an electron like a metal, but it's a gas like a nonmetal, and it can also gain an electron like another nonmetal. Clear as mud, right?
Hydrogen in Everyday Life
Even though it's got this existential element identity crisis, hydrogen is super important in our everyday lives. Water (H2O), for starters. Without it, we'd be in a world of hurt. All those organic molecules that make up, well, everything alive? Loaded with hydrogen.

Fuel cells, the kind that might power your car in the future, use hydrogen to create electricity. Fertilizers that grow our food? Made with hydrogen. Even some industrial processes like making ammonia (another crucial ingredient for fertilizers) rely on hydrogen.
The Verdict?
So, is hydrogen a metal, nonmetal, or metalloid? The honest answer is… yes. It’s a nonmetal in most situations we encounter, but it can become a metal under extreme pressure. It doesn't really fit neatly into any category, which is what makes it so unique and, frankly, a little bit annoying to categorize. It's the element that refuses to be defined. Just like that person at the party, hydrogen marches to the beat of its own atomic drum.
Maybe we shouldn't try to force it into a box. Maybe we should just appreciate its weirdness and the amazing things it does for us.
