Hydrogen Fluoride Intermolecular Forces

Okay, so picture this: I'm in lab, trying to etch some silicon wafers (don't ask), and the guy next to me is wrestling with a bottle labeled "HF." Now, even with gloves and a fume hood, you can practically feel the respect everyone has for this stuff. It's not just about the acid burns (though, yeah, those are a concern). It's this...presence. It's almost like the HF molecules are particularly clingy, you know?
And that got me thinking: what is it about hydrogen fluoride that makes it so… potent, so uniquely hazardous? Sure, it's an acid, but plenty of acids are just, well, acids. HF is different. And the answer, as it often does in chemistry, lies in the intermolecular forces. (Bet you knew I was going to say that, right?)
What are Intermolecular Forces, Anyway?
Basically, intermolecular forces (or IMFs if you're feeling fancy) are the attractions between molecules. They're what hold liquids and solids together. Think of them as tiny, invisible magnets. The stronger the magnets, the harder it is to pull the molecules apart. That's why water boils at a higher temperature than, say, methane. Water has stronger IMFs.
Must Read
There are a few types of IMFs, each with its own level of "stickiness":
- Van der Waals forces (London Dispersion Forces): These are the weakest, but every molecule has them. It's basically the temporary, random clumping of electrons creating temporary dipoles.
- Dipole-dipole interactions: These happen between polar molecules - molecules where one end is slightly positive and the other is slightly negative. Think of it like lining up a bunch of tiny magnets, positive to negative.
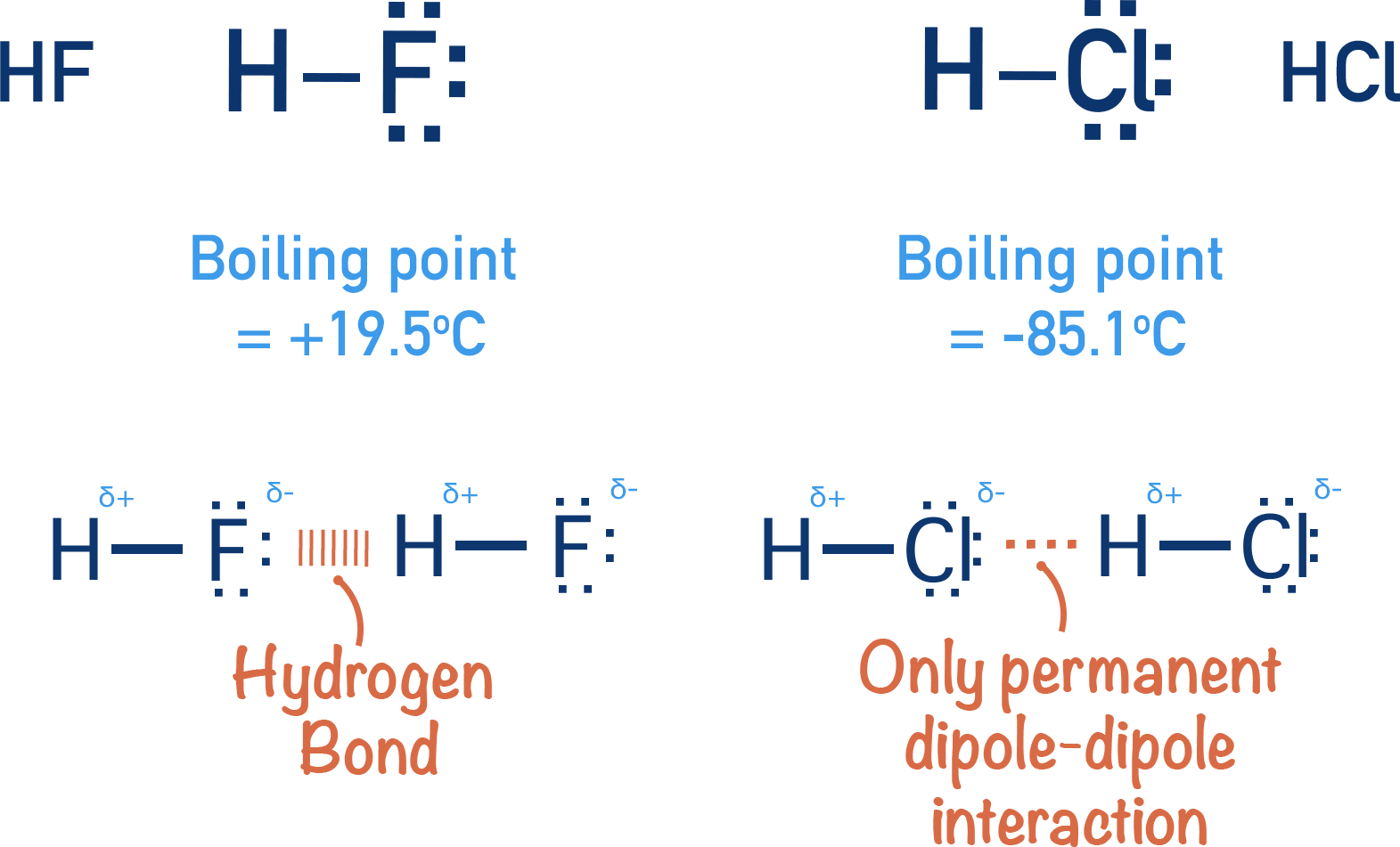
- Hydrogen bonding: This is the big one for HF. It's a special type of dipole-dipole interaction that's much stronger, only happening when you have hydrogen bonded to either fluorine (F), oxygen (O), or nitrogen (N). Coincidence that HF is... well, HF? I think not!
HF: The King (or Queen) of Hydrogen Bonding
So, why is hydrogen bonding so special in HF? Because fluorine is the most electronegative element there is! (Yeah, you probably knew that too from chem class.) That means it really pulls electrons towards itself in the H-F bond. This creates a huge partial negative charge on the fluorine and a large partial positive charge on the hydrogen.

The result? The hydrogen of one HF molecule is strongly attracted to the fluorine of another HF molecule. This forms a strong hydrogen bond. And these hydrogen bonds don't just form between two HF molecules; they form between many of them, creating long, zig-zagging chains or even rings of HF molecules.
Think of it like a bunch of sticky velcro strips all clinging together. Trying to separate those strips takes a lot more energy than separating two smooth surfaces.

Why This Matters (Beyond Etching Silicon)
The strong hydrogen bonding in HF explains several of its unique properties:
- Relatively high boiling point: Compared to other hydrogen halides (HCl, HBr, HI), HF has a surprisingly high boiling point. This is because you need to break those hydrogen bonds to turn liquid HF into a gas.
- Its ability to dissolve ionic compounds that are normally insoluble in water: HF can actually break apart the crystal lattices of some salts because the fluoride ions are very good at stabilizing the positive ions. This is because of…you guessed it…strong interactions, specifically the attraction between F- ions and the positive ions in the lattice.
- Its extreme reactivity: This is where the "danger" comes in. HF's ability to penetrate skin and attack bone (because of the interaction with calcium) is directly related to the strong forces it can exert at the molecular level. Basically, it really, really wants to interact with other stuff.
So, next time you hear about hydrogen fluoride, remember it's not just another acid. It's a molecule with a serious case of "clinginess," all thanks to the power of intermolecular forces, especially the mighty hydrogen bond. And maybe appreciate the people in lab coats who handle this stuff for a living. They’re basically molecular wranglers.
And just a little reminder: Do not try to handle HF at home! Leave it to the professionals with the right equipment and knowledge. It's a fascinating, but dangerous, chemical.