Hydrogen Cyanide Lewis Structure

Alright, buckle up! We're about to dive into the fascinating (and surprisingly dramatic) world of… the Lewis structure of hydrogen cyanide (HCN)! I know, sounds intimidating, right? But trust me, it's less like rocket science and more like a very tiny, very important game of molecular connect-the-dots.
HCN: A Molecular Soap Opera
Think of molecules like tiny actors in a play. Each one has a role, relationships with other actors, and a story to tell. Hydrogen cyanide, HCN, is no different. Our little cast includes hydrogen (H), carbon (C), and nitrogen (N).
Each atom craves a certain number of electrons to be happy and stable. It's like needing a certain number of hugs to feel secure. Hydrogen wants two, carbon needs eight, and nitrogen also wants eight. This neediness is what drives the whole molecular drama!
Must Read
Act 1: The Lone Hydrogen
Poor hydrogen. It’s a bit of a minimalist, content with just two electrons in its outer shell. Hydrogen enters the scene with only one electron to offer. It's like showing up to a potluck with just a single potato chip.
Act 2: Carbon Steps Up
Then there's carbon, the life of the party! Carbon struts in with four electrons, ready to bond with anyone and everyone. Carbon really wants to be the center of attention and achieve that octet of eight electrons!
Carbon needs four more electrons to complete its happy eight. So, carbon is now desperate to connect with any atom that can help him to fulfill the octet rule.
Act 3: Enter Nitrogen, the Intense One
Nitrogen saunters in, radiating intensity. It has five electrons and needs three more to complete its octet. Think of it as a very focused, slightly demanding character.
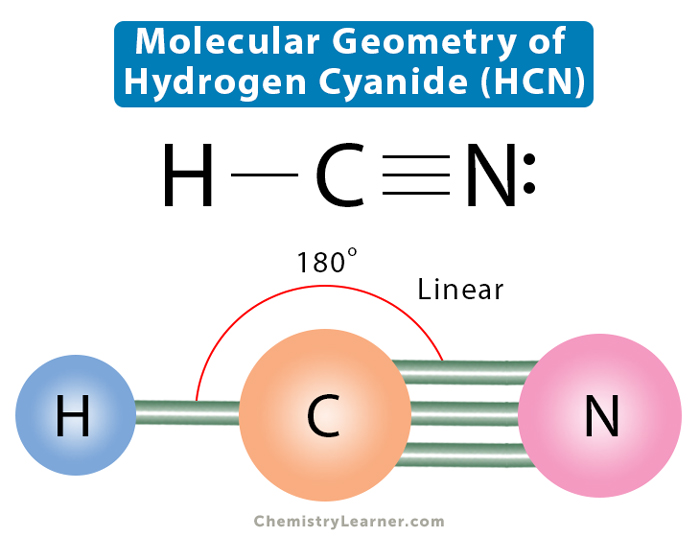
The Big Connection: The Lewis Structure
This is where the fun begins! To draw the Lewis structure, we need to figure out how these atoms can share their electrons to achieve stability. It's all about sharing is caring!
Hydrogen, being the easiest to please, happily forms a single bond with carbon. It shares its one electron with carbon, and carbon shares one back. Hydrogen gets its two electrons, and everyone's happy… almost.
Carbon still needs three more electrons, and nitrogen needs three. What happens next is the equivalent of a dramatic plot twist: a triple bond!
Carbon and nitrogen decide to share three pairs of electrons! It’s like they’re holding hands with three arms each. This is represented by three lines between the C and N atoms in the Lewis structure: C≡N.

Voila! Carbon now has eight electrons (one from hydrogen and three from nitrogen), and nitrogen has eight electrons (three from carbon). Everyone has achieved their octet, and the molecule is stable. It's a chemical happy ending!
Why This Matters (Besides Being Fun)
Okay, so we've drawn a Lewis structure. Big deal, right? Well, understanding the structure of molecules like hydrogen cyanide is incredibly important.
The arrangement of atoms and electrons determines a molecule's properties. This includes how it reacts with other molecules, its shape, and even its toxicity. Knowing the Lewis structure helps scientists predict how a substance will behave.
For example, hydrogen cyanide is highly toxic because it interferes with cellular respiration. The way it binds to certain enzymes is directly related to its molecular structure.
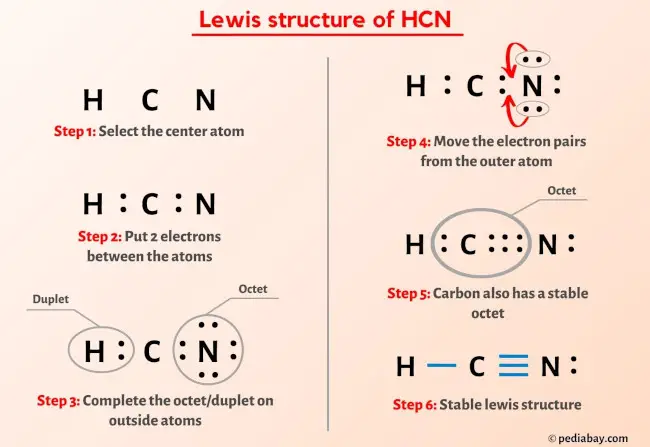
More Than Just Dots and Lines
Think of Lewis structures as a basic language that chemists use to communicate about molecules. It's a shorthand way of representing complex interactions.
While HCN might be associated with danger due to its toxicity, remember that understanding its structure helps us develop antidotes and safer alternatives in various industrial processes.
So, the next time you hear about a complex chemical compound, don't be intimidated! Remember the basic principles of the Lewis structure and think of it as a tiny molecular drama unfolding.
A Little Bit of Humor (Because Why Not?)
Imagine hydrogen cyanide trying to get into a club. Hydrogen, being small, easily gets past the bouncer. Carbon, with its charisma, tries to charm his way in. But nitrogen, intense and demanding, insists on bringing three dates (electrons) with him, causing a bit of a scene at the door.

The bouncer, representing the laws of chemistry, demands that everyone has the correct number of electron "guests" before allowing them in. Only by forming the proper bonds (a single bond for hydrogen, a triple bond between carbon and nitrogen) can they all party inside the club (achieve stability).
Conclusion: Atoms are Just Like Us!
The story of hydrogen cyanide and its Lewis structure shows us that even molecules have needs and desires. They crave stability and seek out ways to achieve it through bonding and sharing.
And just like in any good drama, there are elements of danger, excitement, and ultimately, resolution. Who knew chemistry could be so engaging?
So, go forth and impress your friends with your newfound knowledge of molecular soap operas! You've officially unlocked a deeper level of understanding the amazing world of chemistry.
